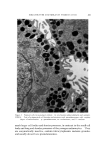
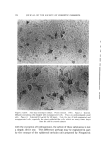
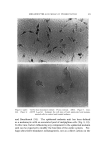
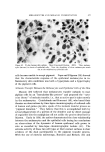
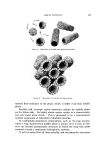
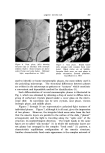
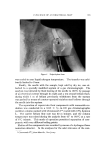
LIQUID CRYSTALS 593 tube as a shampoo. With such a product the hydration to gummy middle phase in use offers interesting possibilities. Middle phase, on the other hand, has been marketed as an auto- mobile soap, as a concentrate for suppliers of liquid soap, and as a shampoo (17). The stiff, elastic isotropic phase-type on the high-moisture side of the middle-phase region of potassium coconut soap has been marketed as a textile soap. The clear, elastic hair-dressings are presumably the same phase type, possibly inverted. Lotions and creams are ordinarily two-phase compositions, and often involve fine crystals rather than mesomorphic phases. Neat/nigre or neat/lye emulsions do exist, however, as in some "liquid detergent" products. Figure 15 shows a representative neat/lye emulsion in the polarizing microscope. ACKNOWLEDGMENT In the preparation of this paper, the author appreciates the valuable help of Messrs. T. J. Flautt, K. D. Lawson, and A. P. Murphy, and of the art and photographic staff at Miami Valley Laboratories. (Received December $, 1967) REFERENCES (1) Friedel, G., Les •tats m•somorphes de la matiare, Ann. Phys. (Paris), 18, 273-474 (1922). (2) McBain, J. W., and Marsden, S.S., The structure types of aqueous systems of surface- active substances and their x-ray diffraction characteristics, Acta Cryst. 1,270-2 (1948). (3) Luzzati, V., Mustacchi, H., and Skoulios, A., The structure of the liquid-crystal phases of some soap and water systems, Discussions Faraday Soc., 25, 43-50 (1958). (4) Clunie, J. S., Corkill, J. M., and Goodman, J. F., The structure of lyotropic mesomor- phic phases, Proc. Roy. Soc. (London), Ser. A., 285,520-33 (1965). (5) Bernal, J. D., and Fankuchen, I., X-ray and crystallographic studies of plant virus preparations, J. Gen. Physiol., 25,111-65 (1941). (6) Hermann, C., Die Symmetriegruppen der amorphen and mesomorphen Phasen, Z. Krist., 79,186-221 (1931). (7) Mabis, A. J., Structure of mesomorphic phases, Acta Cryst. 15, 1152-7 (1962). (8) Doseher, T. M., and Void, R. D., Phase relations in the system: sodium stearate- cetane, J. Colloid Sci., 1,299-312 (1946). (9) Roseyear, F. B., The microscopy of the liquid crystalline neat and middle phases of soaps and synthetic detergents, J. Am. Oil Chemists' Soc., 31,628-39 (1954). (10) McBain, J. W., Lazarus, L. H., and Pitter, A. V., Die Anwendung der Phasenregel auf das Seifensieden, Z. Physik,. Chem., A147, 87-117 (1930). (11) McBain, J. W., and Lee, W. W., Vapor pressure data and phase diagrams for some concentrated soap-water systems above room temperature, Oil &' Soap 20, 17-25 (1943). (12) Vold, R. D., and Vold, M. J., Successive phases in the transition of anhydrous sodium palmitate from crystal to liquid, J. Am. Chem. Soc., 61,808-16 (1939).
JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS (13) Roseyear, F. B., unpublished data. (14) Adapted from Ferguson, R. H., Phase phenomena in commercial soap systems, Oil & Soap, 9, 5-8, 25 (1932). (15) Ralston, A. W., Hoffman, E. J., Hoerr, C. W., and Selby, W. M., Studies on high mo- lecular weight aliphatic amines and their salts, J. Am. Chem. Soc., 621, 1598-1601 (1041). (16) Lutton, E. S., Phase behavior of the dimethyldodecylamine oxide-H20 system, J. Am. Oil Chemists' Soc., 4il, 28-30 (1900). (17) Wood, R. C., to Procter & Gamble Co., ?lastic Detergents and Method of Making Same, U.S. Patent 2,580,713 (January 1, 1952).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)