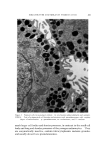
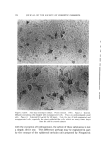
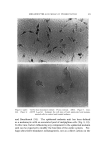
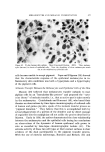
J. Soc. Cosmetic Chemists, 19, 627-640 (Aug. 19, 1968) Studies on Nonaqueous Emulsions ROBERT V. PETERSEN, Ph.D.,* and RICHARD D. HAMILL, Ph.D.'• Presented December 6, 1967, New York City Synopsis--A departure from traditional methods and concepts of emulsion technology has resulted in the development of a family of nonaqueous emulsions which possess a variety of interesting and unique properties. Liquids representing the polar phase include glycerin, propylene glycol, and polyethylene glycol 400. Olive oil was used as the nonpolar phase liquid. Representative anionic, cationic, and nonionic surfactants were employed. Con- ventional theories and methodology are not readily translatable to the nonaqueous systems in many instances. For example, emulsion type and method of preparation seem to be more closely related to the chemical nature of the surfactant than to other types of classification such as the HLB system. INTRODUCTION Modern definitions of the term "emulsion" are broad in scope and include any system in which a liquid is dispersed as droplets through- out a second, immiscible liquid. For example, USP XVII (1) defines an emulsion as "... a two-phase system in which one liquid is dispersed in the form of small globules throughout another liquid." This, and similar definitions impose no limitations on the nature of the liquids which may be involved and designate liquids by name only when referring to specific emulsion types (e.g., oil-in-water or water-in-oil). In contrast to the inclusiveness of the definitions, classical concepts and experimental studies have been restrictive in scope and, almost without exception, have been limited to systems containing water and an oil. Despite the abundant references to studies on the effects of * College of Pharmacy, University of Utah, Salt Lake City, Utah 84112. t Baxter Laboratories, Inc., Morton Grove, Ill. 60053. 627
628 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS surfactants on aqueous and aqueous-oil systems, little is to be found in scientific literature regarding the technology of nonaqueous or nonoil- containing emulsions. Indeed, most investigators cautiously cir- cumvent conditions which would favor emulsion formation. In many industrial and laboratory procedures, such as the extraction of a natural product from its crude source or from another solvent, emulsification must be considered undesirable and a phenomenon to be avoided. The scarcity of information regarding nonaqueous and non-oil- containing emulsions is unfortunate because many unique and useful properties may be found among such preparations. In recognition of the need and desirability of obtaining information on the technology of these systems, investigations on the capacity of various types of surfactants to emulsify glycerin and olive oil (2, 3), and on the physical properties of the emulsions thus formed (4-6), have recently been carried out in this laboratory. These cosmetically elegant emulsions range in physical appearance from clear, transparent, viscous fluids to soft, white creams. Some of them exhibit a remarkable degree of stability at exceedingly low surfactant concentration. In many instances, widely accepted theories regarding the formation and prop- erties of water-oil emulsions cannot be related directly to the non- aqueous systems. Because of the many desirable features of the glycerin-oil emulsions and because other l:olyols and glycols are widely used in cosmetic preparations, it seemed important to broaden the scope of these in- vestigations to include other representative compounds, namely pro- pylene glycol and polyethylene glycol 400. (Subsequent use of the term "polyol" in this report is intended to include these glycols.) The results of these studies and a comparison of the properties of the various types of emulsions serve as a basis for this report. EXPERIMENTAL The methods employed in the emulsification process are similar to those reported in previous publications which described the emulsifying effects of ionic (2) and nonionic (3) surfactants on glycerin and olive oil. Briefly, in Method I the surfactant or surfactant precursor (sa- ponifying amine) was added to the requisite volume of polyol. A quantity of olive oil sufficient to produce the desired phase volume was added in small portions with vigorous trituration after each addition. The quantities of reagents and procedure in Method II were the same except for the order of mixing. In Method II, the surfactant or sur-
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)