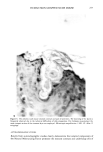
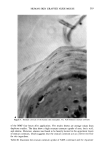
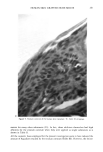
j. Soc. Cosmet. Chem., 37, 249-265 (July/August 1986) The athymic nude mouse grafted with human skin as a model for evaluating the safety and effectiveness of radiolabeled cosmetic ingredients ROBERT V. PETERSEN, Department of Pharmaceutics, College of Pharmacy, Universit 3, of Utah, Salt Lake City, UT 84112,' M. SERPIL KISLALIOGLU, Department of Pharmaceutics, College of Pharmacy, Idaho State University, Pocatello, ID 83209,' WEN-QUAN LIANG, and SEN-MAW FANG, Department of Pharmaceutics, College of Pharmacy, University of Utah, Salt Lake City, UT 84112,' MONSOOR EMAM, Division of Dermatology, College of Medicine, University of Utah, Salt Lake City, UT 84112,' and STEVEN DICKMAN, Microbiological Development and Control, Inc., 520 Wakara Way, University of Utah Research Park, Salt Lake City, UT 84108. Received July 30, 1984. Presented at the Annual Scientific Meeting of the Society of Cosmetic Chemists, New York. December 1-2, 1983. Synopsis The safety and efficacy of cosmetic ingredients have been major concerns for the cosmetic chemist for many years. However, most animal models currently used to evaluate the safety of various topically-applied substances do not provide data which can readily be correlated with anticipated human responses to these substances. Recent studies have demonstrated clearly that a wide variety of chemical agents have the ability to penetrate the skin and even to enter the systemic circulation. Many researchers are studying a number of drugs as potential candidates for use in transdermal delivery systems. It has been shown that some cosmetic ingre- dients may also permeate the skin to varying degrees. This report describes a new technique for deter- mining the extent of penetration of radiolabeled agents into the stratum corneum and underlying tissues. The technique utilizes human skin which has been grafted onto the athymic nude mouse. Penetration of various labeled materials is determined either by tape-stripping the skin and counting radioactivity using a scintillation counter or by obtaining punch biopsies of the grafted skin followed by rapid freezing, cryo- stating, and autoradiography of the biopsied skin tissues. Components of the Natural Moisturizing Factor (NMF) were used in these studies. These and other studies show conclusively that various amino acids, urea, lactic acid, p-aminobenzoic acid, and other agents penetrate into and through the stratum corneum of the grafted human skin to varying degrees. This model likely approximates natural human cutaneous absorption characteristics better than any other non-human model commonly used today. INTRODUCTION Human skin, once considered to be an impervious barrier which protects the body from 249
250 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS invasion by foreign substances, is now acknowledged as a highly complex organ through which many substances can diffuse or be transported. The significance of this fact has become more evident largely through the research efforts of an increasing number of investigators involved in studies on transdermal permeation of various drugs. Many consider this to be the optimal mechanism for the design of drug delivery systems. In such systems the skin may be the rate-limiting factor in others, skin diffu- sion is sufficiently high that synthetic polymers are needed to provide the rate-control- ling function. Several examples of systemically active transdermal drug delivery systems are presently available and many others are under investigation. The research cited above has heightened the awareness of many investigators and regula- tory agencies to the potential advantages as well as risks of exposure to a variety of environmental substances, including those used in cosmetic formulations. The review of over-the-counter (OTC) drug ingredients and claims by the Food and Drug Adminis- tration (FDA), which began in 1972 (1-3), has had a profound impact on the cosmetics industry. The FDA/OTC review has resulted in the removal from the market of ingre- dients used in cosmetic products (e.g., hexachlorophene) or has modified the testing procedures and label claims (e.g., ultraviolet light-absorbing agents used as sunscreens) (4). In response to these actions the Cosmetic Ingredient Review Program (CIR) (5) was established by the cosmetics industry to ascertain the safety, or lack of safety, of ingre- dients used in cosmetic products. There are many problems in evaluating the effectiveness and potential toxicologic prop- erties of topically applied substances. Some of these are: 1) lack of a non-human animal model with skin which closely resembles that of the human, 2) lack of uniformity in skin characteristics between different human subjects and even skin from different sites on a single human subject (6,7), 3) lack of an in vitro model system which approximates closely the in vivo characteristic of human skin, 4) lack of uniformity in sensitivity and allergic reactions to applied substances, and 5) the unacceptability of using human subjects for evaluating potentially toxic or dangerous substances. Recent publications by Elias et al. (6), and Handjani-Vila et al. (7) have demonstrated that structural fea- tures and lipid content of stratum corneum have greater impact on percutaneous trans- port of topically applied substances than does the thickness of this component of the skin. Qualitative variations of transcellular, intracellular, and appendageal transport also represent unresolved questions concerning percutaneous absorption of applied sub- stances. During the mid 1970s, techniques for transplantation of human skin onto the congeni- tally athymic (nude) mouse were reported (8,9) and the transplanted skin shown to retain the histologic features of the donor (10, 11). It was demonstrated that the trans- planted skin would exhibit characteristic growth of epidermal appendages (9, 12). Krueger eta/. (13,14) further demonstrated that pig skin transplanted onto the nude mouse retained its pretransplant properties relative to proliferation and barrier func- tions. In 1981 Krueger and Shelby (15) further substantiated the maintenance of donor qualities of the grafted skin by exposure of the graft to agents which modify epidermal proliferation, e.g., 12-O-tetradecanoyl-phorbol- 13-acetate (TPA). Reifenrath eta/. (16) compared several animal models, including the human skin grafted athymic nude mouse and pig skin grafted athymic nude mouse, for their trans- dermal absorption of nine topically applied radiolabeled compounds. These investi-
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)