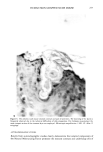
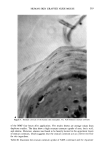
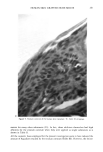
HUMAN SKIN GRAFTED NUDE MOUSE 251 gators concluded that a significant correlation existed between the human skin/nude mouse and human values, whereas no significant correlation existed between human values and those of pig skin/nude mouse or hairless dog values. Due to several factors, including those enumerated above, the availability of the animal model and the expertise required for a highly interdisciplinary study, it was concluded that a study should be conducted to develop the methodology for utilizing the human skin/nude mouse as a model for evaluating the uptake, transport, and retention of selected cosmetic ingredients by the stratum corneum. METHODS GRAFTING OF MICE The mice used in this study were obtained from a colony maintained by Gerald G. Krueger, M.D., Division of Dermatology, University of Utah. The colony was origi- nated by mating pathogen-free female Balb/c mice heterozygous for nude with Balb/c males homozygous for nude (15). Because the animals are athymic they must be main- tained in a pathogen-free environment. Otherwise, standard maintenance procedures were utilized. Details on animal maintenance, skin harvesting, storage, and grafting procedures have been described in previous publications (8-15). The human skin used for transplantation was obtained from females undergoing abdominoplasty. Skin was obtained shortly after surgery and was dermatomed to about 0.4 mm thickness and kept at 2-5øC in RPMI1640 (Flow Labs. Inc., Virginia) to which was added 10% Fetal Bovine serum (FBS) (Flow Labs, Inc.). Skin maintained under these conditions could be used for transplantation for up to five days. The skin samples were cut into square sections approximately 1.5 cm per side. Grafting was accomplished by anesthetizing nude mice about 11- 16 weeks of age with intraperitoneal injection of 97.5 mg/kg of 4% chloral hydrate solution, clipping away a section of the mouse's skin behind the left front leg, placing the human skin over the denuded area, and bandaging the area. Under these conditions vascularization of the graft requires about 3-5 days and healing is complete in about three to four weeks. EXPERIMENTAL TECHNIQUES Autoradiographic analyses. In order to assess the uptake and permeation of the radiola- beled chemicals, autoradiography was chosen as a preliminary study. Employing this technique, the treated sections of skin were first cleansed with a mild aqueous soap solution and cotton swab. Punch biopsies 4 mm in diameter were then taken, placed in OCT Fluid (Tissue Tech. Tissue Tech II OCT Mounting Fluid), and immediately frozen in liquid nitrogen. Frozen samples were cut into thin crossections (about 6 •m) with a cryostat (American Optical Model 975C histostat cryostat) and mounted onto glass slides. The sections were then autoradiographed by standard techniques. Briefly, this includes coating the slide with a special photographic emulsion (Eastman Kodak, Nuclear Track Emulsion NTB-3) allowing several (2-5) days exposure, then devel- oping and fixing the exposed slide. The resulting slides were photomicrographed in
252 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS order to obtain photographic images defining the areas where radioactive substances were located. Measurement of stratum corneum uptake by spreading and stripping. Application methodology. In this technique, following complete healing animals were prepared for transdermal study by first anesthetizing with hexobarbital sodium (90- 100 mg/kg). A specific area of the human skin graft was outlined by means of templates which had been cut to various sizes. Test and control preparations were then administered in the amount of 5-10 }xg/cm 2 and spread uniformly over the marked area. Similar tests were performed on marked sections of the mouse's own skin. Fluid samples were measured by micropipers, whereas viscous fluids or semisolid dosages were measured gravimetrically on a 5-place electronic balance (Merrier, Model AE 163). In order to prevent any disturbance of the treated area, the animals were restrained in standing position by placing the feet through appropriately spaced holes in a plastic device. The test materials used in the experiments were mainly selected components of the Natural Moisturizing Factor (NMF) (Kolmar Laboratories, Inc.). In addition to the investigation of the single components of the Natural Moisturizing Factor (NMF) such as lactic acid, alanine, and urea, a radiolabeled NMF condensate was synthesized in our laboratory using 14C-labeled glutamic acid (New England Nuclear) and an unlabeled sugar. The formulation of the Natural Moisturizing Factor (Aqualizer E J) was reported earlier (17). Its main component is the NMF condensate which is a polymerization product of amino acids like glutamic acid, alanine, and glycine with aldehyde sugars like glucose. The polymerization reaction resulting with NMF condensate is known as the Maillard reaction and can be shown as follows: R - NH 2 3- O = C -- C - R'--- R - N = C - C - R' + H20 I OH OH 14C-labeled glutamic acid was the only labeled component of the condensate. This product had an activity of 0.88 p•Ci/g. However, in order to simulate the commercially available natural moisturizing factor (Aqualizer E J) (Kolmar Laboratories, Inc.), the condensate was further mixed with 14C-labeled glycine, alanine, urea, glucose, and lactic acid (New England Nuclear) to give an activity of 20 p•Ci/g. Controls were prepared in an identical manner except that non-radioactive chemicals were included into the formulation. Preliminarily, both NMF condensate and NMF were tested for stratum corneum uptake from 1% to 10% w/v aqueous solutions. To facilitate uniform spreading, 0.01% w/v sodium dodecyl sulfate (Mallinckrodt, USA) was added to these solutions in a few instances. Later the radiolabeled Aqualizer was included in four different types of cos- metic bases in 1% to 10% w/v ratio to investigate their effects on the penetration of the NMF. The results from 10% concentrations are given in this paper due to low levels of radioactivity in strippings using 1% concentrations. The general formulations of the cosmetic bases used can be summarized as follows: Base A.' This was an w/o-type cream. It contained petrolatum, lanolin, mineral oil, branched chain esters, PEG stearate, polysorbate 60, sorbitan stearate, trierhanoi- amine, carhomer, propylene glycol, and water.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)