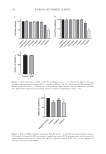
143 MICROENCAPSULATED INSECT REPELLENT METHODS FORMULATION DEVELOPMENT IR3535 was selected as the active ingredient because it has multiple advantages over DEET. Most notably, those advantages include the lack of odor, lack of oily feel on skin, and better toxicological profile. The selected concentration was 20% w/w. In view of decades of expertise by our scientists in topical formulation development, careful consideration was given to the chemical properties for the selected excipients for inclusion within the microencapsulated emulsions. Consideration was given to the selection of and preferences for cosmetically acceptable fluidic carriers (e.g., water, ethanol, glycerol) suspending and emulsifying agents (e.g., sorbitan oleate, polysorbate 20, lecithin, sodium lauryl sulfate) hydrophobic oils or lipids (e.g., isoeicosane, isopropyl myristate, isopropyl palmitate) viscosity agents (e.g., polyamide-3, cetyl alcohol, carrageenan, cellulose, methyl cellulose, polyethylene glycols, or carbomer homopolymer type A) film formers (e.g., dimethicone, acrylates copolymer) preservatives (e.g., phenoxyethanol, ethylhexylglycerin, parabens) stabilizing agents such as antioxidants (e.g., butylated hydroxytoluene, butylated hydroxyanisole, citric acid) and Lewis acids and Lewis bases for microencapsulation of emulsions (e.g., chitosan, algin, xanthan gum). The International Nomenclature of Cosmetic Ingredients served as a guide during selection of excipients. Fragrance was optional. From these possible excipients, a “generic ingredients list” for IR3535-containing microencapsulated formulations was compiled, including the target concentrations of the ingredients. The list was utilized to conceive a formulation referred to as prototype version 1.0 (Table I). As described in the results section, successive derivatives of version 1.0 resulted in formulations 2.0 and 2.55. Table I Generic Ingredients List to Generate Sustained-Release Repellent Formulation Prototype Version 1.0 Category Chemical name or class % (w/w) Active IR3535 20 Emollient Acyclic alkane ∼2 Emulsifier Sorbitan fatty acid ester ∼1 “ Polysorbate 1 Encapsulation Algin + cellulose 1 “ Chitosan 1 “ Xantham gum 1 Thickener Polyamide ∼1 “ Cetyl alcohol 1 “ Carrageenan 1 Film former Silicone ∼ 1 “ Nonsilicone ∼ 1 pH/antioxidant Citric acid (optional) 1 Preservatives Preservatives 1 Fragrance Fragrance (optional) 1 Active + excipients ∼30 water ∼70
144 JOURNAL OF COSMETIC SCIENCE The method of preparation of repellent version 2.0 is the following: • In a heated mixing container, combine the emollient, the emulsifiers, two of the thickeners (polyamide-3 and cetyl alcohol), and the film former dimethicone (Phase A). Commence heat to 90°C. Stir until melted Phase A is completely homogenous. • Discontinue heating, and when temperature is below 80°C, add IR3535 (Phase A1) gradually, stirring to incorporate. • While Phase A (including Phase A1) is cooling, in a separate heated container, prepare Phase C. Add citric acid into the purified water and stir until completely solubilized, then add the trisodium citrate (dihydrate). Once the trisodium citrate is completely solubilized, the pH should be approximately 6.2. Add the final amount of purified water to complete the final weight of Phase C, and begin heating to 50°C. • In a separate container mix the (dry) encapsulation ingredients and the thickener carrageenan (Phase B), then add Phase B into Phase A slowly and gradually, stirring to avoid local freeze-out of gel. Mix thoroughly. • When Phase C is at 50°C, add Phase A/B into Phase C. When all Phase A/B has been added to Phase C, recommence stirring. Shear hard and homogenize. Continue to shear with the homogenizer to form creamy white emulsion. • Add the film former acrylates copolymer (Phase D) and the preservatives (Phase E) into the emulsion sequentially with stirring, while cooling the product. • If required, add additional purified water for appropriate final weight. • Check that the pH of the final emulsion is between 6.0 and 6.5. For composition version 2.0, the target pH is 6.2. • Add (optional) fragrance (Phase F) into the emulsion once the temperature is below 40°C, Mix thoroughly. TESTING OF STABILITY AND PHYSICAL ASPECTS This study focused upon repellent compositions that manifest stabilized formulation properties at RT and/or accelerated temperature at 40°C. The formulations were also stability tested at 50°C (data not shown). Weekly assessments of pH were performed. The linear regressions of the time in weeks to arrive at a reduction in pH loss of 1.0 units were determined. Each formulation was also assessed for viscosity at 25°C using an analogue rotationary viscometer (PCE RVI 1, spindle #4, 12 rpm). The preferred viscosity range for the lotion was 3,000–10,000 cps. The texture and fluid flow pattern of a lotion, as well as a white to off-white color, were also assessed. The concentration of IR3535 within the lotions stored at 40°C and RT were analyzed by gas chromatography. This was conducted by Stillmeadows Inc. (Sugar Land, Texas) using a gas chromatograph Trace™ 1310 with autosampler, Chromeleon™ (Thermo Fisher Scientific, Waltham, Massachusetts) integration software, Phenomenex ZB-FFAP column (30 m length times 0.32 mm inner diameter times 0.5 µm film thickness). The operating conditions included an injector temperature of 220°C injection volume of 1.0 µL split ratio 25 flame ionization detector at 260°C column (oven) temperatures (initial 100°C, hold 1.0 minute, increase 20°C/min, final 250°C, hold 6.5 minutes, total run time 15 minutes) carrier gas hydrogen, 3.0 mL/min constant flow (by electronic flow control) and make-up gas nitrogen. Integration parameters were measurement type area retention
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)