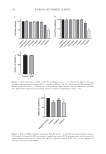
145 MICROENCAPSULATED INSECT REPELLENT time(s) internal standard methyl undecanoate at 4.3 minutes and the active ingredient 3-[n-N-butyl-N-acetyl]-aminopropionic acid ethyl ester at 7.4 minutes. ARM-IN-CAGE MOSQUITO EFFICACY TESTING ON HUMAN SUBJECTS Repellency efficacy tests (arm-in-cage) were conducted for version 2.0 and prototype version 1.0 against the mosquito species Aedes aegypti at Laboratorios Ecolyzer LTDA (Sao Paulo, Brazil). These tests were performed according to the OPPTS 810.3700 methodology (July 7, 2010), which is one of a series of test guidelines established by the Office of Chemical Safety and Pollution Prevention of the EPA, for use in testing pesticides and chemical substances to develop data for submission to the agency. The arm-in-cage procedure for testing of LivFul Inc.’s (Cheshire, United Kingdom) microencapsulated emulsions of 20% IR3535 was approved (May 22, 2018) by the Institutional Review Board for experiments on human subjects. Mosquitos. There were host-seeking females of A aegypti 55 mosquitos per cage (1 mosquito/1,160 cc) in acrylic cages 40 × 40 × 40 cm (64,000 cc). Number of subjects. There were 10 subjects. The same volunteer subjects were used as an untreated group (to validate the study and as a mosquito attractiveness control) and as treated volunteer subjects with the repellent lotion group using a different arm. An attractiveness test to the participants was performed before the application of the repellent product at each time point. This procedure was performed to verify whether the participants were suitable for the study. At least five landings/bites were needed to be recorded for acceptance. Dose applied. The dose was 1 g lotion/600 cm2 of skin (forearm). The area of the forearm of each participant was calculated according to the formula {[(M1 + M2 + M3 + M4 /4) × C]/600}, where M1 was the forearm circumference at wrist height (cm), M2 and M3 were two equidistant measurements of the forearm circumference between elbow and wrist (cm), M4 was the forearm circumference at the elbow, and C was the forearm length (cm). Methodology. Before each evaluation with the forearm treated with repellent, mosquito activity evaluations were performed in all test cages. The exposure time of the forearm without treatment was 1 minute inside the cage or until 5 landings or bites. The treated group exposed their arm for 5 minutes starting after 30 minutes from the application of the product and every hour until evidence of first bite. The CPT is the number of minutes (or hours) elapsed between topical application and the first landing and/or probing (biting) of the insect. CFB is the second incidence of an insect probing (biting). IN VITRO ANTI-INFECTIVE TESTING To determine whether versions 1.0 and/or 2.0 have anti-infective properties, the bacteriological standard test methodology BS EN 1276:2019 was used at Melbec Microbiology Ltd. (Haslingden, United Kingdom). In brief, BS EN 1276:2019 uses a quantitative suspension of Pseudomonas aeruginosa (ATCC 15442), Escherichia coli (ATCC 10536 and/or K12 NCTC 10538), Staphylococcus aureus (ATCC 6538), and Enterococcus hirae (ATCC 10541), with an acceptance criterion of a 5 log10 reduction in viable bacterial counts. An aliquot of the lotion was directly added to a series of test suspensions of P aeruginosa, E coli, S aureus, and E hirae respectively. The suspension was mixed with bovine albumin
146 JOURNAL OF COSMETIC SCIENCE (0.3 g/L), which was used as the interfering substance for the test conditions. After 60 seconds contact time, a neutraliser was immediately added to stop the effects of the disinfectant, and a sample of the corresponding mixture was poured into a bacterial medium plate and incubated. After incubation, the number of surviving bacteria was counted and compared against the original culture number. A 5 log (base 10) reduction within all 4 species met the formally established standard, although reductions below this level and/or in less than 4 species may (also) be indicative of antimicrobial activity. RESULTS FORMULATION DEVELOPMENT When the lotion prototype version 1.0—containing 20% w/w IR3535 and according to the generic ingredients list of Table I—was subjected to stability testing at RT and accelerated temperatures (e.g., 40°C and/or 50°C), it manifested rapid reductions from the initial pH over the course of weeks to months. The reductions in pH by 1.0 units occurred within 5.1 weeks at 40°C and 16.4 weeks at RT. A variance of pH of 1.0 units or greater within this predetermined time (i.e., 12 months at RT) was deemed as an undesirable property it may be a key factor used as a determinant of the imputed or formal shelf life. The pH drift underscores that some component(s) of the prototype version 1.0 were undergoing chemical hydrolysis or degradation, as evidenced by increased concentration of hydronium ions (H3O+) or protons, detected by a pH meter. Lower pH and/or the presence of degradants within the formulations might be disadvantageous to human or animal skin. Note that none of the initial formulation variants, including version 1.0, contained trisodium citrate, although some variants contained citric acid as an optional acidifier/antioxidant. According to the manufacturer of IR3535 (Merck KGaA), the ideal pH for IR3535 is between 6.0 and 6.5. A challenge of using IR3535 as the selected active ingredient in these encapsulated repellent formulations is that in order to adequately solubilize, suspend, or emulsify the IR3535 and the other essential excipients, a pH lower than its inherent ideal pH of IR3535 can be desirable. For human skin, a pH of approximately 5.0–6.5 is appropriate, and approximately 5.5 is often preferable or optimal in topical consumer products. These two parameters are in apparent opposition logic suggests that a low pH might be desirable in the process of producing an IR3535-containing encapsulated formulation, but a higher pH (approximately 5.0–6.5) is desirable for application of the formulation to human skin to avoid unnecessary skin irritation. It follows that the manufacturing process steps (e.g., sequence of addition of the ingredients, temperatures, and mixing intensity) in preparing the formulations may also impact ingredient solubilization, suspension, emulsification, and encapsulation in a pH-dependent manner. Thus, empirical formulation development and analyses via a reiterative process can improve from an initial prototype formulation toward preferred formulations. In multiple efforts to ameliorate the reductions in pH and concentration of the active ingredient, attempts were made to stabilize the formulation(s) with: (a) addition of NaOH to increase the initial pH (b) addition of NaOH to restore (increase) the pH after the formulation had manifested some degree of pH loss and (c) addition of an antioxidant, with the intent of retarding the chemical instability. None of these three empirical approaches
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)