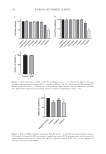
147 MICROENCAPSULATED INSECT REPELLENT adequately addressed the chemical instability issue(s). The instability persisted even when NaOH or an antioxidant was included. It is likely obvious to an experienced formulation scientist that the back neutralization of a citric acid–containing solution (or emulsion) with sodium hydroxide is inherently equivalent to the use of a sodium citrate (or hydrates thereof) buffer solution, differing only in order of addition of the component parts. Nevertheless, this approach did not resolve the issue of pH getting lower over time. The concentration of IR3535 within prototype version 1.0 was assayed at various time points using gas chromatography. The analysis revealed that the degradation of IR3535 at 24 months at 20°C exceeded the WHO and EPA guidelines’ allowed tolerances of 5% to 6% for repellent products, with a declared nominal content of active ingredient of 10% to 25% w/w or w/v and stored at 20°C ± 2. These findings (as previously mentioned) further underscored the problems and confounding variables to be addressed. In summary, the problematic issues faced during formulation development included: (a) most notably rapid pH loss (b) loss of IR3535 (c) a preference for a lower pH for solubility, suspension, emulsification, and encapsulation during formulation development (d) preference for human skin of a pH of approximately 5.0–6.5, and more preferably 5.5 and (e) attempts to maintain appropriate pH conditions by the addition of NaOH or an antioxidant not being sufficient. Thus, multiple attributes of the excipient components, concentrations, and sequence of addition (order) were evaluated to optimize the desired formulation. To prevent the rapid pH reduction manifested by prototype version 1.0 during stability testing (see following section), a formulation version 2.0 was prepared that was essentially prototype version 1.0 but that included trisodium citrate dihydrate (2.024% w/w) plus citric acid (0.095% w/w). This buffered formulation contained a total of 30.63% active ingredient plus excipients, with an initial pH of 6.2. Formulations with two chemical versions of sodium citrate were studied in the dihydrate form and the monohydrate form. To maintain equimolar citrate ion concentrations, less of the monohydrate was required relative to the dihydrate. The dihydrate form was routinely used in commercial consumer products and foods, was less expensive, and may be preferable for the formulation(s). Formulation variants were also prepared with a concentration range of sodium citrate– citric acid to identify preferred amounts of the buffer. Concentrations ranged from 0.2% to 5.0%. In general, the optimal concentration for chemical stability and physical aspects was approximately 2%. Formulation version 2.55 was identical to version 2.0, except it contained 1.80% trisodium citrate dihydrate: 0.31% citric acid, at initial pH 5.5, and 69.38% water. TESTING OF STABILITY AND PHYSICAL ASPECTS A major goal during formulation development was to limit the reduction in pH to no more than 1.0 units within 12 months (and preferably longer) at RT. This desirable increased pH stability was also expected to stabilize the concentration of IR3535, so that it did not exceed 5% to 6% loss over the imputed or formal shelf life. The slopes of the linear regressions (with r2 values 0.9) were extrapolated into imputed (estimated) weeks to reach a pH loss of 1.0 units—thus, from an initial pH of 6.2 downward to 5.2 for version 2.0, or from an initial pH of 5.5 downward to 4.5 for version 2.55. Table II reveals that linear regression analyses of weekly measurements of the unbuffered repellent lotion prototype version 1.0 manifested rapid reductions in pH by 1.0 units within
148 JOURNAL OF COSMETIC SCIENCE 5.1 weeks at 40°C and 16.4 weeks at RT. These times were deemed to be undesirable. However, at 40°C, the linear regression of the slope revealed that the buffered version 2.0 (two variants Bd2 and Bi2) at initial pH of 6.2 reduced the rate of pH drift by 5.3 times and 5.5 times, respectively. At 40°C the buffered version 2.55 (20a) reduced the rate of pH drift by 17.7 times. At RT, the buffered versions 2.0 (Bd2 and Bi2) reduced the rate of pH drift by 5.8 times and 4.2 times, respectively. Also, at RT, the buffered version 2.55 (20a) reduced the rate of pH drift by 23.5 times. If the time to arrive at a loss of 1.0 pH unit is selected as one of the criteria for establishing the imputed or formal shelf life, then the sodium citrate:citric acid buffered versions 2.0 (Bd2 and Bi2) can last for at least 1 year prior to a reduction in pH of 1.0 units. Furthermore, version 2.55 (20a) with an initial pH of 5.5 is expected to be stable for multiple years. These results indicate that chemical stability, of which pH is an important marker, has been substantially enhanced by the selected compositions, and especially so at an initial buffered pH of 5.5. The shelf-life estimates may also be established as a function of the concentration of the active ingredient (e.g., IR3535), but the variance should not exceed 5% or 6% loss of the active ingredient over the duration of the imputed or formal shelf life. The concentration of IR3535 of version 2.0 was assessed at 26 weeks at 40°C and RT and was determined to conform to the acceptable variance. Thus, the sodium citrate–citric acid buffered formulation 2.0 (initial pH of 6.2) stabilized not only the pH but also the active ingredient from hydrolysis/degradation. Surprisingly, as previously noted, the formation of a citric acid buffer system by the addition of sodium hydroxide to a citric acid–containing solution did not stabilize the pH the formulations formed by inclusion of trisodium citrate (or the hydrates thereof) were effectively stabilized. EFFICACY OF FORMULATIONS ON HUMANS AGAINST MOSQUITOS IN LABORATORY ARM-IN-CAGE STUDIES The repellent efficacy against mosquitos was assessed for buffered version 2.0 and the unbuffered prototype version 1.0. A laboratory efficacy test (arm-in-cage) was conducted for the version 2.0 on 10 human participants against the mosquito species A aegypti. The dose was 1.0 g of repellent lotion per 600 cm2 applied to the forearm. Table III demonstrates that none of the participants experienced a CFB (i.e., second bite) prior to 13 hours. At 13 hours into the study, 4 of the 10 participants experienced mosquitos landing and/or biting. Thus, the CPT for the protection against the mosquito species Table II Chemical Stability: Elapsed Time (Weeks) to Reach a pH Reduction of 1.0 Unita Version 1.0 - 2.0 (Bd2) 2.0 (Bi2) 2.55 (20a) Buffered pH na 6.2 6.2 5.5 Weeks @ 40°C 5.1 27.2 27.8 90.1 Weeks @ RT 16.4 94.3 68.5 384.6 a Version 1.0 is the unbuffered prototype version 2.0 (Bd2) has ∼2% sodium citrate:citric acid buffer with sodium citrate monohydrate version 2.0 (Bi2) is ∼2% sodium citrate:citric acid buffer with sodium citrate dihydrate version 2.55 (20a) is ∼2% sodium citrate:citric acid buffer and with sodium citrate dihydrate.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)