153 MICROENCAPSULATED INSECT REPELLENT (4) D.R. Barnard, U.R. Bernier, K.H. Posey, and R.D. Xue, Repellency of IR3535, KBR3023, para- menthane-3,8-diol, and deet to black salt marsh mosquitoes (Diptera: Culicidae) in the Everglades National Park, J. Med. Entomol., 39(6), 895–899 (2002). (5) J.E. Cilek, J.L. Petersen, and C.E. Hallmon, Comparative efficacy of IR3535 and deet as repellents against adult Aedes aegypti and Culex quinquefasciatus, J. Am. Mosq. Control Assoc., 20(3), 299 –304 (2004). (6) U. Thavara, A. Tawatsin, J. Chompoosri, W. Suwonkerd, U.R. Chansang, and P. Asavadachanukorn, Laboratory and field evaluations of the insect repellent 3535 (ethyl butylacetylaminopropionate) and deet against mosquito vectors in Thailand, J. Am. Mosq. Control Assoc., 17(3), 190–195 (2001). (7) S.P. Frances, D.O. MacKenzie, K.L. Rowcliffe, and S.K. Corcoran, Comparative field evaluation of repellent formulations containing deet and IR3535 against mosquitoes in Queensland, Australia, J. Am. Mosq. Control Assoc., 25(4), 511–513 (2009). (8) S. Licciardi, J.P. Herve, F. Darriet, J.M. Hougard, and V. Corbel, Lethal and behavioural effects of three synthetic repellents (DEET, IR3535 and KBR 3023) on Aedes aegypti mosquitoes in laboratory assays, Med. Vet. Entomol., 20(3), 288–293 (2006). (9) S.K. Nakayama, R. Graf, A.M. Bohlmann, J. Zurlage, H. Epstein, M. Ranzani, and F. Pflüecker, Influence of sex ratio and density on the comparative repellency of IR3535® against caged Aedes, Anopheles, and Culex mosquitoes, J. Cosmetic Sci., 70(4), 197–207 (2019). (10) C. Costantini, A. Badolo, and E. Ilboudo-Sanogo, Field evaluation of the efficacy and persistence of insect repellents DEET, IR3535, and KBR 3023 against Anopheles gambiae complex and other Afrotropical vector mosquitoes, Trans. R. Soc. Trop. Med. Hyg., 98(11), 644–652 (2004). (11) M.M.N. Reynoso, E.A. Seccacini, J.A. Calcagno, E.N. Zerba, and R.A. Alzogaray, Toxicity, repellency and flushing out in Triatoma infestans (Hemiptera: Reduviidae) exposed to the repellents DEET and IR3535, Peer J, 5, e3292 (2017). (12) R.A. Alzogaray, Behavioral and toxicological responses of Rhodnius prolixus (Hemiptera: Reduviidae) to the insect repellents DEET and IR3535, J. Med. Entomol., 53(2), 387–393 (2016). (13) J.F. Carroll, J.P. Benante, M. Kramer, K.H. Lohmeyer, and K. Lawrence, Formulations of deet, picaridin, and IR3535 applied to skin repel nymphs of the lone star tick (Acari: Ixodidae) for 12 hours, J. Med. Entomol., 47(4), 699–704 (2010). (14) T.J. Naucke, S. Lorentz, and H.W. Grünewald, Laboratory testing of the insect repellents IR3535 and DEET against Phlebotomus mascittii and P. duboscqi (Diptera: Psychodidae), Int. J. Med. Microbiol., 296 Suppl 40, 230–232 (2006). (15) E.N.I. Weeks, G. Wasserberg, J.L. Logan, J. Agneessens, S.A. Stewart, and S. Dewhirst, Efficacy of the insect repellent IR3535 on the sand fly Phlebotomus papatasi in human volunteers, J. Vector Ecol., 44(2), 290–292 (2019). (16) Manual on development and use of FAO and WHO specifications for pesticides, First Edition – third revision, World Health Organization, (2016). (17) K. Rossington, The Science of Chlorine-Based Disinfectant 2014, accessed June 23, 2022, https://www. manufacturingchemist.com/news/article_page/The_science_of_chlorine-based_disinfectant/103112. (18) S. Tienungoon, P. Pinnil, K.-O. Kreelieng, and S. Onket, Quantitative bactericidal efficacy of alcohol- based handrub in Thailand using modified EN 1276: 2009 method, Bulletin Dept. Med. Sciences (Thailand), 61(2), 60–72 (2019). (19) A. Şahiner, E. Halat, and E.A. Yapar, Comparison of bactericidal and fungicidal efficacy of antiseptic formulations according to EN 13727 and EN 13624 standards, Turk. J. Med. Sci., 49(5), 1564–1567 (2019). (20) S. Messager, K.A. Hammer, C.F. Carson, and T.V. Riley, Assessment of the antibacterial activity of tea tree oil using the European EN 1276 and EN 12054 standard suspension tests, J. Hosp. Infect., 59(2), 113–125 (2005). (21) Z. Iyigundogdu, S. Kalayci, A.B. Asutay, and F. Sahin, Determination of antimicrobial and antiviral properties of IR3535, Mol. Biol. Rep., 46(2), 1819–1824 (2019). (22) S.P. Carroll, Prolonged efficacy of IR3535 repellents against mosquitos and blacklegged ticks in North America, J. Med. Entomol., 45(4), 706–714 (2008).
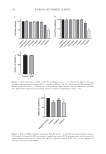
154 J. Cosmet. Sci., 73, 154–165 (May/June 2022) Address all correspondence to Quan An, anquan@ynbyjk.com The Antipigmentation and Antioxidant Activity of Sanchi Flower Extract JUAN WANG, PEIYU WANG, XUE SHAO, TONG HUO, JITAO LIU, QIANGHUA QUAN AND QUAN AN Yunnan Baiyao Group Health Products Co., Ltd., Kunming, Yunnan, P.R. China (J.W., P.W., X.S., T.H., J.L., Q.Q., Q.A.) East Asia Skin Health Research Center, Beijing, P.R. China (J.W., P.W., X.S., T.H., Q.Q., Q.A.) REAL DermaSci & Biotech Co., Ltd., Beijing, P.R. China (J.W., P.W., T.H., Q.Q., Q.A.) Yunnan Baiyao Group Shanghai Science & Technology Co., Ltd., Shanghai, P.R. China (J.L., Q.A.) Accepted for publication June 20, 2022. Synopsis Long-term ultraviolet (UV) irradiation stimulates melanogenesis by direct effects on melanocytes and by indirect effects on keratinocytes releasing melanogenic factors. Hyperpigmentation is a major cosmetic concern. Thus, melanin synthesis inhibitors are popularly used as brightening agents in cosmetics. It has been reported that Sanchi flower extract (SFE) exerts various biological effects including hypotensive, hemostatic, and anti-aging activities. The authors explored the potential of SFE as a cosmetic ingredient. On the one hand, it could significantly inhibit melanin production both in B16-F0 cells and in the three-dimensional skin model. In addition, intracellular tyrosinase activity decreased after treatment with SFE. On the other hand, it upregulated cellular glutathione peroxidase activity and total antioxidant capacity in HaCaT cells irradiated with UVB radiation in contrast to the model group. These results indicated that SFE showed obvious antipigmentation potency and alleviated oxidative stress caused by UVB. INTRODUCTION Sanchi, as a famous and precious Chinese medicine, has been commonly used throughout history as an alternative remedy. It has been used for medicinal purposes to promote blood circulation, relieve swelling and pain, and counteract blood stasis (1). Its various parts— including roots, flowers, and leaves—are used for producing medicinal formulations. The main active ingredients of Sanchi are composed of saponins, amino acids (2), flavonoids, and polyphenols, which display strong antioxidant activity and anti-inflammatory potency (3). However, the antimelanogenesis and skin-protective effects of Sanchi flower extract (SFE) are rarely reported.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)