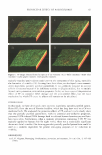
2 JOURNAL OF COSMETIC SCIENCE Taurine, 2-aminoethanesulfonic acid (Figure 1), is one of the most important organic osmolytes. In the skin, taurine is highly concentrated in the granular and upper spinous layer of the epidermis (5.5 µmol/g epidermal tissue) (5). The levels in isolated stratum corneum are very low ( 0.07 µmol/g stratum corneum) (5). Recently, it has been shown that epidermal keratinocytes accumulate taurine when the skin is exposed to a dry environment (6). Moreover, in vitro studies revealed that the expression of the taurine transporter and taurine uptake is also stimulated by UV irradiation of keratinocytes (7). In addition to cell volume regulation, many other functions have been described for taurine, including (i) cell membrane stabilization (8), (ii) stabilization of proteins (9), and (iii) protection form UV-induced apoptosis ( 6). It is interesting to note that taurine accumulation is induced by environmental factors that also disturb the epidermal barrier function to water loss. Barrier function is main- tained by extracellular lipid membranes located in the stratum corneum (10). The major barrier lipids are ceramides, cholesterol, and fatty acids that form multilammelar struc- tures with low water permeability in a 1: 1: 1 molar ratio (11). Epidermal barrier function is a highly dynamic system. Exposure to a dry environment or perturbation of barrier function stimulates the synthesis and secretion of barrier lipids by the outermost granu- lar keratinocytes (12-14). Topical application of anionic surfactants such as sodium dodecyl sulfate (SDS) increases transepidermal water loss (TEWL) and induces epidermal inflammation and hyperpro- liferation (15). Epidermal interleukin-lalpha (IL-lalpha) is a major proinflammatory cytokine involved in the initiation of inflammation and hyperproliferation after SDS- induced barrier disruption (16-18). IL-lalpha can induce other cytokines such as IL-6, IL-8, granulocyte/macrophage colony-stimulating factor, and intercellular adhesion mol- ecule-1 (19), and is expressed in the whole epidermis when epidermal hyperplasia is induced by repeated barrier disruption (20). Barrier lipid synthesis and accumulation of taurine are both stimulated by epidermal stress factors associated with barrier perturbation and inflammation. It is unknown, however, whether these two responses reflect independent cutaneous mechanisms to cope with stress factors and water loss or whether they interact with each other. Therefore, we studied the role of taurine in SDS-induced barrier perturbation. MATERIALS AND METHODS MATERIALS Taurine was obtained from Ajinomoto (Tokyo, Japan). An o/w-emulsion was used as a vehicle containing 2.00% (wt/wt) glyceryl stearate (Cognis, Duesselorf, Germany), 0.75% arachidyl glucoside (Seppic, Paris, France), 9.00% dibutyl adipate (Cognis), Figure 1. Chemical structure of taurine (2-aminoethanesulfonic acid).
TAURINE AND PREVENTION OF DRY SKIN 3 1.00% Dow Corning 9040 fluid (Dow Corning, Midland, Texas), 1.50% behenyl al- cohol (Seppic), 2.75% arachidyl alcohol (Seppic), 1.00% cetearyl alcohol (Cognis), 0.30% carbomer (Goldschmidt, Essen, Germany), 1. 70% aluminium starch octenyl succinate (National Starch, Bridgewater, New Jersey), 1.00% Simugel NS (Seppic), 2.50% glycerol (Cognis), 2.00% propylene glycol (Brenntag, Muehlheim, Germany), and 5 .00% 1,6-hexandiol (BASF, Ludwigshafen, Germany). IN VITRO ASSAY FOR SURFACTANT-INDUCED SKIN IRRITATION Reconstructed epidermis (EpiDerm-200) was obtained from MatTek (Ashland, USA) 18 days after airlift and incubated according to the manufacturer's instructions. In addition, tissues were topically incubated with 5 µl of 0.18% (wt/wt) SDS in water for 1 h. After rinsing with phosphate-buffered saline, 2 mg/cm2 of the vehicle containing 1 % taurine or the vehicle alone (both at pH 5 .2) were applied for 24 h. Incubations with demin- eralized water or an emulsion containing 0.1 % (wt/wt) of the glucocorticoid triamcino- lone acetonide (Kortikoid-ratiopharm® Creme) were performed as negative or positive controls, respectively. Cellular viability was quantified using the MTT-test (MTT, 3-[ 4,5-dimethylthiazol-2-yl}-2,5-diphenyltetrazoliumbromid) (Sigma, Deisenhofen, Germany) as described previously (21). Lactate dehydrogenase activity in the culture medium was measured using a test kit obtained from Roche (Mannheim, Germany) according to the manufacturer's instructions. Quantification of IL-lalpha and PGE2 was performed using test kits available from R&D Systems (Minneapolis, Minnesota), with dilution of the culture medium at 1:4 and 1:10, respectively. LIPID ANALYSIS AND HISTOLOGY Reconstructed epidermis was obtained from SkinEthic (Nice, France) 1 7 days after airlift and incubated according to the manufacturer's instructions. Tissues were harvested and lipids were extracted according to Wertz et al. (22) in each of three solvent mixtures (chloroform/methanol 1/2 [v/v} chloroform/methanol/water 1/2/0.5 [v/v/v} and chlo- roform/methanol 2/1 [v/v }). Lipid extracts were applied to HPTLC-plates (Merck, Darmstadt, Germany) and separated using automated multiple development (Camag, Berlin, Germany), based on 15 steps with decreasing polarity, using appropriate mix- tures of methanol, chloroform, diisopropylether, and n-hexane. For quantitative analyti- cal HPTLC determination, increasing amounts of standard lipids (N-stearoyl- sphingosine, cholesterol, oleic acid, cholsterol sulfate, cholesterol oleate, phosphatidyl- ethanolamine, and phosphatidylcholine, all obtained from Sigma) were applied. After development, plates were air-dried, sprayed with 8% (w/v) H 3 PO4 containing 19% (w/v) CuSO4 , and charred at 180°C for 10 min, and the lipids were quantified by photodensitometry (Camag, Berlin, Germany). For light microscopy, samples were fixed by immersion in phosphate-buffered paraformaldehyde ( 4% ), embedded in paraffin, sectioned (5 µm), and stained with hematoxylin and eosin. EXPERIMENTALLY INDUCED SKIN BARRIER PERTURBATION A total of 78 volunteers with normal skin were included in the study. At first, 100 µl of an aqueous solution of 0.75% sodium dodecyl sulfate (SDS) was applied to the forearm
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)