IODOPHOR-IODINE SHAMPOOS* By A. CANTOR, PH.D., S. MosT, PH.D.,t and M. V. SHELANSK•, M.D. Industrial Toxicology Laboratories, Philadelphia, Pa. IT •s THE purpose of this paper to provide some guidance and orientation for the cosmetic chemist, with reference to the development of iodophor-iodine products. Iodine belongs to the group of elements known as halogens chlorine, fluorine and bromine are other familiar members. It occurs only in traces in most natural substances, but is especially concentrated in sea waters• marine plants and shell fish. Iodine is essential for the normal functioning of mammals, fishes, other animals and perhaps plants. In addition, iodine and its compounds have many interesting actions which have made it important in pharmacology, bacteriology and chemistry. Iodine exists in three major states: (1) as elemental iodine--here would be included solutions and tinctures of iodine and some complexes with iodine (2) as iodide ion--as in sea water and the blood stream of man and (3) as organically bound iodine, such as the thyroid hormone and iodinated proteins, in addition to special pharmaceutical organically bound iodine products, of either natural or synthetic origin (1). Elemental iodine is an extensively used germicide because of its activity against micro/Srganisms. It is unique in its nonselectivity for bacteria, viruses, fungi and other inhabitants of the microscopic world. However, it does not distinguish between bacterial and mammalian proteins, and so in recent years attempts have been made to harness the bactericidal powers of the element while controlling its toxicity. Acute poisoning is rare in spite of the occasional suicide attempts with tincture of iodine deaths have been reported from 2-3 gin. of iodine, with recovery from as much as 10 gin. (2). An occasional individual may show a marked sensitivity to local application, and a small number of fatal cases due to this sensitivity have been reported (3). Compared to the number of individuals who have used iodine in some form or other, this is admittedly a very small percentage. Iodism is somewhat more common * Presented at the May 18, 1956, Meeting, New York City. t West Laboratories, Long Island City, N.Y. 419
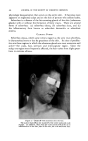
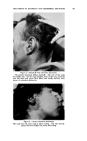
420 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS it is usually associated with regular oral ingestion of iodide although there is much individual variation, it usually occurs with moderate to large doses of iodides used for a length of time, and is usually manifested by irritative phenomena of the skin and mucous membranes. Iodophor-iodine complexes are iodine in combination with a carrier. The carrier is a compound which both solubilizes the iodine, and carries it into solution on dilution with water. The complexing of iodine with a carrier results in (1) the reduction of iodine vapor pressure to very low levels (2) the reduction of toxicity as measured by eye or skin application, and in many cases by oral administration (3) changes in certain iodine adsorption equilibria and chemical reaction rates (4). The carrier may be a surface active agent (cationic, anionic or nonionic), or it may be a water-soluble polymer, such as PVP. Carriers differ in ihe quantity of iodine they carry, the effectiveness with which they do so, their stability, their chemical reactiveness with iodine (the amount of iodine they consume by chemical combination), their toxicity, and their ability to be formulated into useful and practical preparations. The total iodine of iodophor-iodine preparations consists essentially of a physically complexed but chemically available (titratable) fraction, and one which is chemically bound and is neither available nor titratable. It is essentially the titrat- able iodine which is germicidal. Iodophor-iodine preparations possess the well-recognized germicidal and disinfectant properties of free iodine, with less toxicity and irritancy than is usually associated with the free element. The first carrier investigated was the water soluble polymer, PVP (5). The second surface active or detergent carrier studied and described was the ethylene oxide condensate of Co alkyl phenol (6). This is a nonionic surface active agent and was shown to be an efficient carrier of iodine. Since then many other carriers have been studied, and much has been learned of their characteristics and usefulness. The most useful and stable carriers were the nonionic surface active agents. Surfactant iodophors have evoked much interest and have found uses as detergent-sanitizers, germicides and disinfectants (6-11). The fungicidal and bactericidal properties of iodine, the diminished toxicity and irritancy of iodophors and the effects of iodine on keratin and other fibers suggested their introduction into the field of cosmetics, initially as a shampoo. Iodine is capable of being adsorbed onto many protein fibers, such as silk, wool and hair. Adsorption of iodine onto fibers is greatly accelerated at acid pH values, in contrast to iodination which proceeds more rapidly in alkaline media. The amount of iodine adsorbed by silk fiber from a number of iodine solutions was studied by Rice and Beal (12) who found that ad- sorption from a triethanolamine hydriodide solution is considerably greater
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)