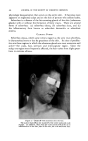
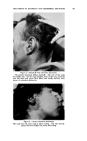
IODOPHORE-IODINE SHA/vlPOOS 425 oral toxicity values of the various types of nonionic surfactants, though as a general class they are less toxic than the anionics or cationics. There are differences among the members of the same series. A comparison of values for low and high molecular weight surfactants of the same series generally shows a decrease in toxicity as the molecular weight is increased. There are differences in the iodine toxicity of the iodophor-iodine preparations at different ratios of carrier to iodine, depending in part upon the efficiency of the carrier as an iodine complexing agent. For formulating a shampoo which will be acceptable from the standpoint of eye safety, it is necessary in each case to determine the maximum safe iodine content for a specified carrier or iodophor. For example, the Pluronic and Igepal series of surfactants were examined closely. Certain lower members of the series have LD50 rat values ranging from about 2.5 gm./kg. to 7 gm./kg. When complexed with iodine, the LD:,0 values likewise cover a threefold range, at another level. In light of the above range of toxicities for the detergents themselves, it follows that the various members of the series are dissimilar in their iodine complexing nature, as reflected by oral toxicity. A like observation was made when the materials were studied in rabbit eyes. The order of eye irritancy does not necessarily follow the order of oral toxicity. Such a correlation did exist in one, but not in the other, of the two series of nonionic surfactants re- ferred to above. These conclusions were true for both 20:1 and a 100:1 ratio of carrier to iodine, i.e., a 5 per cent iodine solution and a 1 per cent iodine solution, in the surfactant carriers, as well as for other intermediate ratios and aqueous dilutions. The compatibility of mixtures of judiciously chosen members of these two series of nonionics, permits of a convenient flexibility in formulation, with reference to the ultimate toxicity and per- formance of the product. The hazard of generalization, with reference to the toxicity of these iodophor-iodine complexes, is further illustrated by a third type of nonionic surfactant. In this case, the oral toxicity was markedly lower than that of either the Pluronic or Igepal members, and eye irritancy was higher. The toxicologic examination of very high members of the Pluronic series, as iodophors, is particularly interesting, since the acute oral toxicity for such complexes approaches the toxicity which characterizes PVP - Iodine at the same 10:1 ratio. A general observation may be made with reference to the importance of the ratio of iodophor to iodine. If the product under consideration is to be applied topically, whether to the hair or otherwise, the detoxifying function of the iodophor may be minimized if the iodophor is overloaded with iodine. This becomes strikingly apparent when a comparison is made of, e.g., 20:1 ratios of iodophor to iodine, as compared with higher ratios, for the nonionic surfactants. As the ratio is increased, the limiting
426 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS factor in the toxicity (oral, eye or skin) becomes the toxicity of the iodophor itself. It follows that maximum safe iodine concentrations to be used in a shampoo are a function of the intrinsic eye irritancy of the iodophor carrier itself and of the ratio of the carrier to the iodine. By way of illustration, for the three series of nonionic surfactants referred to above, when a 20:1 ratio of iodophor to iodine is used, the maximum safe (rabbit eye test) concentration of iodine in the finished shampoo, would range from 0.5 to 5 per cent (i.e., from a 10 per cent solution in water to the undiluted iodophorAodine complex), depending upon which member within the particular surfactant series was used as the carrier. In addition to the variation among the members of a given series, the Pluronic surfactant- iodine complexes as a group have been found characterized by a lower eye irritancy than the other two nonionic series studied, and the difference is increased as the ratio of carrier to iodine is increased. It is of interest to estimate the quantity of iodine which may be absorbed through the skin as a result of the use of an iodine shampoo. An iodophor- iodine shampoo formulation containing 0.5 per cent titratable iodine is germicidal and fungicidal as a prophylactic shampoo. For the purpose of calculation, if one uses a properly formulated 1.5 per cent iodine shampoo, and a double shampooing is used, 10 mi. of the shampoo, or 150 mgm. of iodine, are applied to the hair and scalp. Approximately the same quan- tity of iodine is used in painting a 5-inch square patch of skin with tincture of iodine. However, it is obvious that an insignificant part of the iodine in the applied shampoo reaches the skin, since the iodine colored lather can be rinsed away without residual color, and since the scalp remains un- stained. If one premises that one per cent of the applied iodine is available for absorption through the scalp, and if skin absorption of iodine for humans is as inefficient as for rabbits, 1 per cent of the iodine available for absorp- tion may be absorbed. That is, less than 100 gamma, and probably closer to 10 gamma may be absorbed, if any. The latter quantity is far less than the required daily iodine intake, far less than the quantity consumed in iodized salt, and less than the iodides present in six oysters or a glass of Italian wine (30). REFERENCES (1) Salter, W. T., Karandikar, G., and Block, P., 7. ?lm. Pharm. ?lssoc., Sci. Ed., 38, 626 (1949). (2) Soilmann, T., "A Manual of Pharmacology and Its Applications to Therapeutics and Toxicology," Philadelphia, W. B. Saunders Company (1948), p. 816. (3) Seymour, W. V., Jr., ?lrch. Internal Med., 59, 952 (1937). (4) Terry, D. H., and Shelanski, H. A., Proc. 38th Meeting Chem. Specialties Mfg. Assoc., Dec. (1951). (5) Shelanski, H. A., U.S. Patent Application Serial No. 135,519 and U.S. Patent No. 2,739,922. (6) Cantor, A., and Shelanski, H. A., Soap Sanit. Chemicals 27, 133 (1951). (7) Cantor, A., and Shelanski, H. A., Proc. 38th Meeting Chem. Specialties Mfg. Assoc., Dec. (1951).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)