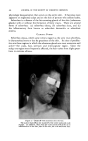
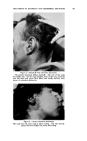
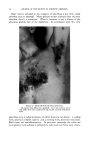
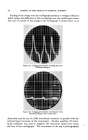
460 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Even though the combination of selenium sulfide with bentonite had been .a definite advance, it was still of limited usefulness as is indicated by the fact that no satisfactory commercial preparation was developed during the life of the Nordlander patent. During this time the problem was further investigated by Baldwin and Young, and a formulation of broad usefulness came only with the discovery that the selenium sulfide- bentonite mixture should be combined under certain conditions in sus- pension with a rather high concentration of a suitable detergent of low sensitizing properties (3). It is this combination of essential ingredients which is now marketed under the trademark Selsun © . CHEMICAL NATURE OF SELENIUM SULFIDE Before discussing this product further, it is of interest to look into the chemical nature of selenium sulfide. Because it is prepared by the fusion of selenium and sulfur in a molar ratio of 1: 2 it is frequently called selenium disulfide. However, our studies identified no component with the structure SeS2 present in the material (4). An x-ray diffraction pattern shows clearly the presence of crystalline selenium monosulfide, SeS. All of the remaining material consists of solid solutions of selenium and sulfur in an amorphous form. As amorphous selenium and amorphous sulfur may both exist as octamers (Sea and S8), it is reasonable to conclude that at least part of the selenium disulfide melt is in the form Se•S,•, where n q- m = 8. No new crystalline complex was formed when selenium disulfide was fused with bentonire. CLINICAL STUDIES The first extensive controlled clinical studies with Selsun suspension were conducted by Slinger and Hubbard (5) and by Slepyan (6). They found the preparation active in controlling seborrheic dermatitis of the scalp of all degrees of severity, 85 to 95 per cent effective in controlling the mild form commonly known as dandruff. These findings have subse- quently been confirmed in many thousands of cases so that, as has been suggested (7), the preparation probably deserves to be considered the therapeutic standard as an antidandruff agent. However, investigations with selenium sulfide have now been expanded sufficiently to establish that its effectiveness is not limited to the scalp. Seborrheic dermatoses of other areas of the glabrous skin are also controlled with a selenium sulfide jelly, for example those of the external ear canal, the nasolabial fold and the eyelids. Selenium sulfide must be used with considerable caution on the eyelids, because of irritation if the material is allowed to get in the eye, but the precautions necessary are justified by the relief produced in the condition known as B/ep/•aritis margina/is, a granulation of the eyelids which is very frequently associated in the patient with dandruff (8, 9).
SELENIUM SULFIDE AS AN ANTIDANDRUFF AGENT 461 There is also good evidence that selenium sulfide may be combined with other therapeutic agents in formulations offering the additive benefits of each drug. ToxicITY Some dermatologists have been reluctant to prescribe a selenium sulfide preparation because of the toxic properties of selenium. A cautious attitude is well justified, although it seems clear that there is an ample margin of safety for use under medical supervision. Slinger and Hubbard (6) have reported the toxicity studies on Selsun suspension carried out in the Kettering Laboratory in the Department of Preventive Medicine and Industrial Health, College of Medicine, University of Cincinnati. The suspension was used in the recommended manner in 19 patients with frankly diseased scalps. Urine selenium determinations were made on samples collected during the twenty-four hours following application of the medica- tion. No significant increment in selenium excretion could be demon- strated in the subjects studied. Two patients with very severe atopic dermatitis, in whom large areas of the scalp were denuded and fissured, were treated daily with the preparation for nineteen days in one case and fourteen days in the other. The first patient showed no trend in selenium excretion while the second showed an equivocal trend toward an increase in excretion. Flesch (10) has suggested that urinary excretion alone may not be an adequate criterion of the safety of a selenium preparation, re- ferring to two cases of poisoning in patients with a history of daily exposure to selenium for over thirty years and in whom low urinary selenium levels were found (11, 12). Nevertheless, none of the subjects studied for possi- ble absorption of selenium showed a sustained urinary concentration of selenium above levels found in normal healthy persons whose exposure to selenium is limited to contact with that present in the general food supply. The chief reason for the safety of selenium sulfide is, of course, its in- solubility. Safety can be assured, however, only by a very strict specifi- cation which limits soluble selenium compounds in the product to a very low figure. As Selsun suspension is labeled plainly as a poison, it is to be expected that attempts would be made to commit suicide by swallowing a large amount of it. Such attempts have been made, all unsuccessful. Fortunately, the large amount of detergent in the preparation helps to defeat the would-be suicide by making the product a fairly effective emetic. Us•. Ao^I•rST To the extent that the cause of dandruff is unknown, the means by which Selsun suspension produces its effect is likely also to be unknown. Nevertheless, it is worthwhile to correlate a number of interesting facts about Selsun to the various theories on the etiology of dandruff.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)