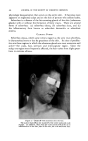
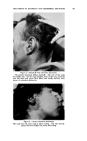
SELENIUM SULFIDE AS AN ANTIDANDRUFF AGENT 459 (29) Combes, F. C., "Colloidal Sulphur, Some Pharmacodynamic Considerations and Their Therapeutic Applications in 'Seborrheic Dermatoses,'" N.Y. State 7. Med., 46, 401 (1946). (30) Finnerud, C. W., and Riddell, J. M., Jr., "Polythionic Acid in the Therapy of Acne Vulgaris and Seborrheic Dermatitis," drch. Dermatol. and Syphilol., 63, 373 (1951). (31) Wright, C. S., Samitz, M. H., and Brown, H., "Vitamin B6 (Pyridoxine) in Dermatology," [bid., 47, 651 (1943). (32) Forman, J., "A Safe and Efficient Topical Medication," Ohio State Med. St., 51, 987 (1955). (33) Effersoe, H., "The Effect of Topical Application of Pyridoxine Ointment on the Rate of Sebaceous Secretion in Patients with Seborrheic Dermatitis," dcta Dermato-I/enereol., 34, 272 (1954). (34) Botwright, W. E., "Quaternary Ammonium Compounds in Cosmetic and Related Prod- ucts," 5 t. Soc. Cosmetic Chem., 3, 118 (1952). (35) Lesser, M. A., Drug and CosmeticInd., 70, 3 (1952). (36) Ball, F. I., "A New Treatment for Seborrheic Dermatitis," drch. Dermatol., 71, 696 (1955). SELENIUM SULFIDE AS AN ANTIDANDRUFF AGENT* By EDWARD J. MATSON dbbott Laboratories, North Chicago, IlL FOR MANY YEARS chemists concerned with therapeutic agents for dermatologic and other conditions have looked with interest at the element selenium, because of its position directly below sulfur in the periodic table. The possibility that selenium sulfide might be an effective agent was suggested as a by-product of research in quite a different field. Nord- lander at General Electric Company was concerned with developing a sensitive agent which would detect leaks in mercury vapor turbines. Selenium sulfide turned out to be the answer. He showed that the product prepared by heating together at 225øC., one mole of selenium and two moles of sulfur was chemically reactive and yet relatively stable to light and heat (1). It occurred to Nordlander that such an active combination might be useful in dermatology. It was apparent that a wettable preparation was to be desired for such purposes. Nordlander finally showed that the preferred composition was a 50:50 mixture of selenium sulfide and ben- tonite, prepared by heating together the two components at a temperature above the fusion point of selenium sulfide. After cooling, the resultant mass was ground to a fine powder and was then readily suspendible in an aqueous vehicle. Various forms of this composition were tested and were found undeniably effective in certain skin conditions (2). *Presented at the December 15, 1955, Meeting, New York City.
SELENIUM SULFIDE AS AN ANTIDANDRUFF AGENT 459 (29) Combes, F. C., "Colloidal Sulphur, Some Pharmacodynamic Considerations and Their Therapeutic Applications in 'Seborrheic Dermatoses,'" N.Y. State 7. Med., 46, 401 (1946). (30) Finnerud, C. W., and Riddell, J. M., Jr., "Polythionic Acid in the Therapy of Acne Vulgaris and Seborrheic Dermatitis," drch. Dermatol. and Syphilol., 63, 373 (1951). (31) Wright, C. S., Samitz, M. H., and Brown, H., "Vitamin B6 (Pyridoxine) in Dermatology," [bid., 47, 651 (1943). (32) Forman, J., "A Safe and Efficient Topical Medication," Ohio State Med. St., 51, 987 (1955). (33) Effersoe, H., "The Effect of Topical Application of Pyridoxine Ointment on the Rate of Sebaceous Secretion in Patients with Seborrheic Dermatitis," dcta Dermato-I/enereol., 34, 272 (1954). (34) Botwright, W. E., "Quaternary Ammonium Compounds in Cosmetic and Related Prod- ucts," 5 t. Soc. Cosmetic Chem., 3, 118 (1952). (35) Lesser, M. A., Drug and CosmeticInd., 70, 3 (1952). (36) Ball, F. I., "A New Treatment for Seborrheic Dermatitis," drch. Dermatol., 71, 696 (1955). SELENIUM SULFIDE AS AN ANTIDANDRUFF AGENT* By EDWARD J. MATSON dbbott Laboratories, North Chicago, IlL FOR MANY YEARS chemists concerned with therapeutic agents for dermatologic and other conditions have looked with interest at the element selenium, because of its position directly below sulfur in the periodic table. The possibility that selenium sulfide might be an effective agent was suggested as a by-product of research in quite a different field. Nord- lander at General Electric Company was concerned with developing a sensitive agent which would detect leaks in mercury vapor turbines. Selenium sulfide turned out to be the answer. He showed that the product prepared by heating together at 225øC., one mole of selenium and two moles of sulfur was chemically reactive and yet relatively stable to light and heat (1). It occurred to Nordlander that such an active combination might be useful in dermatology. It was apparent that a wettable preparation was to be desired for such purposes. Nordlander finally showed that the preferred composition was a 50:50 mixture of selenium sulfide and ben- tonite, prepared by heating together the two components at a temperature above the fusion point of selenium sulfide. After cooling, the resultant mass was ground to a fine powder and was then readily suspendible in an aqueous vehicle. Various forms of this composition were tested and were found undeniably effective in certain skin conditions (2). *Presented at the December 15, 1955, Meeting, New York City.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)