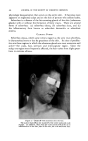
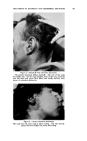
MATERIALS IN THIOGLYCOLATE HAIRWAVING PREPARATIONS 481 6. The extremely unstable character of most certified colors in thio- glycolate solutions, an instability that is likewise aggravated by many perfume materials. 7. The reactivity and the volatility of ammonia, often used in hair- waving preparations its reactivity with numerous perfume materials makes their use difficult if not impossible and its volatility presents tre- mendous difficulties to the perfumer seeking to give a pleasant topnote to the solution. 8. The large number of variables that must be considered in formulating such perfume oils, thus making it almost mandatory that each product be made to order for the particular thioglycolate solution. While it is true that all cosmetics present infinite variety of formulations, the changes in formula normally do not affect the behavior of perfume materials as critically as in a hair wave. Variables that must be taken into account in- clude, but are not limited to, the nature of the thioglycolate salt, the presence of color and the dye used to obtain such color, the nature of the clouding agent (if any), the type of thioglycolate used (whether distilled or ex- tracted) and the nature of the emulsifier or solubilizer used for the perfume. The reactivity of thioglycolic acid has been studied by many workers. The acid readily reacts with aldehydes and ketones, according to Gershon and Rieger (1), to form mercaptals and mercaptoles. As long ago as 1888, Bongartz (2) pointed out that thioglycolic acid combines with benzalde- hyde upon several hours standing, and under slightly exothermic condi- tions, whereas the acid combines with cinnamic aidehyde upon several minutes standing, and with a considerable release of heat. Stricks and Kolthoff (3) showed that the acid requires only a small amount of such metals as copper, manganese and iron to be oxidized in the presence of air to the dithiodiglycolic acid. Inasmuch as these metals are frequently pres- ent in some perfume materials as impurities, such a reaction may be caused by the perfume. With the above in mind, a series of extensive experiments was under- taken in our laboratories, to observe the behavior of individual perfume materials in various thioglycolate solutions and to observe their odor strength, stability of odor, effect on the stability of various clouding agents, effect on both clear and tinted solutions, shelf-life and oven-life and effective- ness of odor coverage when used on the hair. In addition, studies were made of fragrances blended from the various materials to determine similar behavior in proportions closer to those actually used in practice and to ob- serve what has been called by Klarmann (4) "synergisms and antagonisms of groups of perfume, materials." Some 200 different perfume raw materials and mixtures were studied, and the observations are recorded in the following tables. The observations of effectiveness in covering the thioglycolate odor are a consensus of the
482 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS TABLE 1--BEhAViOR OF PERFUME RAW MATERIALS IN CLEAR AMMONIUM THIOGLYCOLATE SOLUTION (No COLOR ADDED) Effect on Odor Odor Se.para- Materials Clarity Coverage Character tlon Discoloration At lco ho ls Alcohol C-10 Clouds Good True None None Benzyl alcohol Clear Fair True None Slight Cinnamic alcohol Clear Fair True None Slight Citronellol Clear Good True None None Geraniol Clear Good True None None Linalool Clear Excellent True None None Phenyl ethyl alcohol Clear Poor True None None Atldehydes Aldehyde C-10 Slight clouding Fair Weakens None None Aldehyde C-11 Slight clouding Fair Changes None Slight Aldehyde C-12 Lauric Clear Good True None None Aldehyde C-12 MNA Slight clouding Excellent True None None Amyl cinnamic alde- ß hyde Clear Good True None None Citronellal Clear Excellent Changes None None Heliotropin Clear Fair True None None Hydroxycitronellal Clear Poor Fair None None Vanillin Clear Fair Thins out None Orange Esters Amyl salicylate Slight clouding Good Fair None None Benzyl acetate Clear Fair True None None Cedryl acetate Clouds Good Fair Slight None Diethyl phthalate Clear None None None None Ethyl phenyl acetate Clear Poor Changes None None Methyl phenyl acetate Slight clouding Fair True None None Phenyl ethyl acetate Clear Fair Changes None Slight Styralyl acetate Clear Poor Fair None None Terpiny! acetate Cloudy Good Fair None None Trichlorostyralyl acetate Clear Good True None Yellows Other Chemical Compounds yellow yellow yellow Bois de rose oil Clear Excellent True None None Cedarwood oil Slight clouding Poor Poor-fair None None Cinnamon oil Ceylon Clear Good Changes None Slight yellow Geranium oil Bourbon Clear Excellent True None None Guaiacwood oil Clouds Strong Changes None None Guaiacwood concrete Clear Poor Changes Insoluble Yellows Labdanum Clear Fair Fair None Yellows Oakmoss Clear Fair True None Dark brown Orange oil cold-pressed Slight clouding Fair Thins out None Slight yellow Patchouli oil Clear Good True None Pink Sandalwood oil Clear Faint True None Slight yellow Vetivert oil Bourbon Clear Good True None None Ylang ylang oil Clear Poor True None None Essential Oils and Other Natural Materials Bromstyrol Slight clouding Excellent True None None Coumarin Clear Poor Changed None None Diphenyl methane Clouds Good Fair None None Diphenyl oxide Clouds Good True None None Eugenol Clear Good Fair None Slight yellow Methyl ionone Clear Good True None Slight yellow Musk xylol Slight clouding Fair Poor Insoluble None Phenyl acetic acid Clear Depresses Thins out None None odor of thio Styrene Clear Good True None Yellows
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)