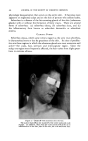
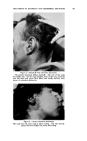
462 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS The infection theory for dandruff is dignified by a long history. The repeated isolation of organisms of the Pityrosporum ovale group from cases of seborrheic dermatitis has been the basis of the assertion that there is an etiologic relationship between these organisms and the disease. In recent years the theory has gained little support and, in fact, it has suffered in the face of data such as those of Spoor and his co-workers (13). These in- vestigators cultured organisms obtained from the scalp of subjects both with and without symptoms of seborrheic dermatitis. They found that 60 per cent of all subjects, diseased or not, harbored Pityrosporum ovale or- ganisms, so that a causal relationship may be doubted. Whether the infection theory is correct or not, it is true that Selsun suspension has antimicrobial activity. Spoor and his co-workers (7) found a 2.5 per cent selenium sulfide suspension to be among the most active agents tested in inhibiting the growth of Micrococ. aureus and albus and Pityrosporum ovale. Our own studies (14) have shown Selsun suspen- sion to have good antifungal activity. The following is a list of several organisms and the dilution of Selsun suspension effective in inhibiting their growth as determined by an agar dilution procedure: Candida albicans .......................... 1:640 Trichophyton mentagrophytes .............. 1:1280 Trichophyton tonsurans .................... 1: 280 Aspergillus niger .......................... 1: 640 Microsporum sp ........................... 1:320 Chaetomium globosum ..................... 1:2560 Myrothecium verrucaria ................... 1:320 Pityrosporum ovale ........................ 1: 320 Nordlander assumed in his patent on the selenium sulfide-bentonite combination (2) that selenium served only as an activator, and that the therapeutically active element was sulfur. This view has been restated by Shair (15), who pointed out several chemical facts of possible signifi- cance. Selenium is a less reactive chemical than sulfur, yet it tremen- dously increases the activity of the latter element. In the photoelectric cell the addition of selenium greatly increases the photosensitivity of sulfur. Physiologically it is possible that selenium, in selenium sulfide, acts as an intramolecular catalyst, replacing part of the sulfur in an S8 molecule and making the sulfur more effective in interfering with the processes causing seborrhea. SKIN EFFECT In the dermatologic armamentarium of drugs, sulfur has been classed as a dermal drying agent. That is, it tends to restrict excretion of skin fats. But Slinger (16) Bereston (17) and others have reported that some seborrheic dermatitis patients treated with Selsun suspension show an
SELENIUM SULFIDE AS AN ANTIDANDRUFF AGENT 463 apparent increase of skin fat about the third or fourth day after treatment with selenium sulfide suspension. No other simple agent administered systemically or locally is known which seems to cause a frank increase in skin excretion of fat. Slinger (16) has made a preliminary report of studies on such patients. Using the techniques developed by Jones, Spencer and Sanchez (18), oil samples collected from the scalp were spread on water to give a mono- molecular film and the quantity was determined by measuring the area of the film. Each sample was analyzed for fatty acids, free and combined, the triglycerides and the waxes. There was a suggestion that the selenium sulfide suspension had pro- duced an increased excretion of skin fat and that the composition had been altered. However, it was more likely that the apparent increase in total skin fat excretion represented the usual quantity of skin fat observed in more fluid form because of a lack of drying action following the applica- tion of selenium sulfide. As Slinger pointed out, it is known that small amounts of selenium will decrease the drying properties of certain drying oils. On the basis of the limited amount of data so far available, it is therefore not possible to state categorically that the action of selenium sulfide is due to intrinsic properties of the combination or to sulfur strongly catalyzed by selenium. On the normal scalp, as on the skin of the whole body, an imperceptible process of desquamation constantly occurs. The horny layer of the epidermis is continually shed as a fine dust, at a rate which just matches the formation of new epidermis from the lower layers of the skin. In dandruff, however, the desquamation process is abnormal in a number of respects (19). Instead of being submicroscopic the scales are visible. . They appear in much greater amount because the proliferation of cells is accelerated. Furthermore, the scales are not mature epithelial keratin but show several indications of being immature. Dandruff scales contain remnants of nuclear structures, they have a higher water content, and they show a higher sulfhydryl content than normal epidermis. The--SH content of the various layers of the skin has recently been re- viewed in a series of studies by Van Scott and Flesch and their co-workers. It was found (20) that all layers of the skin contain some --SH groups with the content of the epidermis less than in the deeper layers. The disulfide content of the epidermis was no greater than that of the deep Malpighian layer. Conversion of--SH to--SS-- therefore does not seem to occur above this depth. Sulfhydryl groups in the upper layers of the skin must there- fore become enclosed within the protein molecule or cross linked by hy- drogen bonding and thus become unreactive. Van Scott and Lyon (21) had shown earlier that soaps and detergents
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)