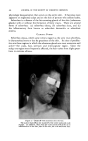
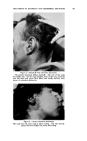
IODOPHORE-IODINE SHAMPOOS 421 than from a tincture of iodine or a Lugol's solution of the same iodine con- tent. Silk fibers can take up approximately 1/1o of their weight of iodine from certain solutions. The adsorption of iodine onto animal fur and skin has been studied in connection with the lustering and curling of furs (13). Iodine was adsorbed by the amino acids of the animal hair more readily from an iodine-iodide solution (Lugol's) than from solutions of iodine in alcohol or other organic solvents. When wool fibers are steeped in an iodine solution, they rapidly assume a dark brown color while the solution becomes correspondingly weaker in iodine. Haller and Holl (14) consider that two almost simultaneous effects take place: first, iodine is taken up by adsorption, and secondly, part of the adsorbed iodine enters into fixed chemical combination with the wool. From a comparison of the tyrosine content of wool and the amount of iodine which would be required to convert all of the tyrosine into diiodo- tyrosine, Blackburn and Phillips (15) concluded that the iodine taken up by wool is probably exclusively substituted on the tyrosine groups to give 3: 5 diiodotyrosine. The rate of removal of adsorbed iodine from iodinated wool is greatest when alkaline detergents are used, and least in acidified solutions. Iodination of wool is said to increase its resistance to shrinkage, and attack by moths, alkalies and other deleterious influences (16). Human hair, in common with wool and a number of other fibers, adsorbs iodine from solutions. We have found the effect of iodine on the hair to depend upon many factors: the type and color of hair, the iodine solution being employed (iodine concentration, presence of other components), the pH of the solution, the temperature, the manner in which it is employed, the time of contact and other factors. Occasional cases have been re- ported of women who dyed their graying hairs a red-brown with tincture of iodine. If one examines Lugol's solution iodine-stained hairs micro- scopically one sees an even cortical coloration little color is lost on stand- ing, or with water washings, but it can be readily removed by alkalinization. Hairs which have been treated with Lugol's solution and then with thiogly- collate preparations show more damage, as seen microscopically and as measured by the elongation and tensile strength of the hair, than do hairs which have not been treated with Lugol's solution. However we have found iodophor and iodophor-iodine formulations more amenable to control in their action on the hair than are the iodine- iodide Lugol's preparation. Less iodine is adsorbed onto hair from such solutions than from Lugol's (at the same iodine concentration and pH) as with Lugol's iodine solutions, more iodine is adsorbed from iodophors at acid pH than at neutral or alkaline pH values. Microscopically, such hairs appear normal and undamaged. It is possible to formulate iodophor- iodine solutions which can be used to treat the hair, to the extent of color-
422 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS ing the hair with adsorbed iodine, and to remove this adsorbed iodine, by rinsing, after the desired contact time has elapsed. This is not similarly practical with Lugol's solution. The swelling and softening effects of cold wave lotions, on iodophor-iodine treated hairs, are no more pronounced than on noniodinated control hairs. There was evidence that the iodophor- iodine treated hairs are in fact less readily damaged by subsequent higher pH's and long contact with cold wave preparations. We have found different batches of hair to differ considerably in their adsorption of iodine. White human hair will adsorb more than blond, brown or black hair white rabbit hairs take up more than white dog hairs. At room tempera- ture, and acid pH, 0.5 gin. of white human hair adsorbed about 80 mgm. of iodine from a 1.5 percent Lugol's iodine solution, about 5 mgm. of iodine from an iodophor-iodine formulation and about 3 mgm. of iodine from a PVP-iodine solution (contact time of fifteen minutes). The effects of iodine on the macro-, micro-, and measurable character- istics of the hair, when such factors as pH, temperature and iodine content are kept equal, therefore depend on the type of iodine solution. Germicidal and fungicidal preparations for use on the hair and scalp have employed as their active ingredients the following substances: (1) quaternary ammonium compounds, (2) actamet or hexacholorophene (G-11), (3) the unsaturated fatty acids such as undecylenic acid, (4) sulfur- containing compounds, (5) metal-containing compounds, such as salts and compounds of copper, mercury or silver, (6) phenolic compounds and derivatives, such as hexylresorcinol, salicylanilide and chlorosalicylanilide, (7) naphthol and quinoline compounds, or (8) halogen-containing com- pounds. This by no means completes the list the multitude and diversity of agents employed or suggested reflects the problems of treatment, and the ineffectiveness or complications in use of the present agents. Quaternary ammonium compounds are substantive to and are strongly adsorbed by many surfaces, including skin and hair since many of them show germicidal and fungicidal activity their use has been suggested in hair formulations (17, 18). However, when tested in vivo, alkyl-dimethyl- benzylammonium chloride (Zephiran) and hexadecylpyridium chloride (Ceepryn) were found to be fungistatic rather than fungicidal, and to be rather ineffective chemically (19). The dyes, such as carbolfuchsin (Castellani's paint) and gentian violet are fungistatic in vitro (20) and have the disadvantage of deeply staining the skin and clothes. It has been reported that most of the so-called fungicidal agents are relatively ineffective in killing the fungus in infected hair and are considered fungistatic (21, 22). The only exceptions to this, according to the literature cited, were tincture of iodine and 90 per cent alcohol. The most prevalent mycotic infections of the hair and scalp are those caused by Microsporon and Trichophyton they cause ringworm of the
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)