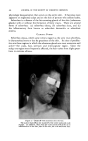
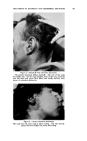
480 JOURNAl. OF THE SOCIETY OF COSMETIC CHEMISTS With the achievement of a six-step process in 1956, I think it fair to say that this is the simplest process ever offered in our industry. Certainly, when it is contrasted with the typical process shown in Fig. 1, evidence of technical progress is apparent. The Prom 1956 method is surely a dif- ferent way of producing a permanent waving result. Perhaps the two procedures might be likened to a train ride in contrast with a plane ride. Either means of transportation will take our customers to the destination they desire. Many women will prefer the simpler, slower, but more cer- tain Prom approach while many others will continue to practice the more complex, but faster chemical oxidation methods. REFERENCES (1) Reed, R. E., DenBeste, M., and Humoiler, F. L., •. $oc. Cosmetic Chem., 1,109 (1948). (2) Brown, A. E., U.S. Patent No. 2,688,972. (3) DenBeste, M., and Reed, R. E., U.S. Patent No. 2,$40,980. (4) Hopkins, R., and Reed, R. E., U.S. Patent No. 2,682,272. (5) Kolb, J. J., and Toennies, G., .qnal. Chem., 24, 1165 (1952). THE BEHAVIOR OF PERFUME MATERIALS IN THIOGLYCOLATE HAIRWAVING PREPARATIONS* By EDWARD SAGARiN and MARVIN BALSAM Standard •lromatics, Inc., lYew York 3, N.Y. FEW COSMETIC preparations present problems so challenging to the perfumer as does the thioglycolate-based cold permanent wave. The sources of the difficulties facing the perfumer are numerous: 1. The strong odor of the thioglycolate salts, or of the impurities therein, requiring powerful odorants to give adequate coverage. 2. The strong odors that are liberated upon the reaction of the thio- glycolate salt with keratin and the subsequent rupture of the disulfide linkages in the hair. 3. The alkaline nature of the medium, excluding from use some organic materials generally used in perfumery, but unstable at a pH of 9 or over. 4. The reactive nature of thioglycolates resulting in the formation of reaction products with certain common constituents of perfumes and the powerful obnoxious odor, even in trace quantities, of some of these reaction products. 5. The relatively unstable nature of most thioglycolate emulsions, their tendency to separate or to cream, which is almost invariably aggravated by many perfume raw materials. * Presented at the May 18, 1956, Meeting, New York City.
480 JOURNAl. OF THE SOCIETY OF COSMETIC CHEMISTS With the achievement of a six-step process in 1956, I think it fair to say that this is the simplest process ever offered in our industry. Certainly, when it is contrasted with the typical process shown in Fig. 1, evidence of technical progress is apparent. The Prom 1956 method is surely a dif- ferent way of producing a permanent waving result. Perhaps the two procedures might be likened to a train ride in contrast with a plane ride. Either means of transportation will take our customers to the destination they desire. Many women will prefer the simpler, slower, but more cer- tain Prom approach while many others will continue to practice the more complex, but faster chemical oxidation methods. REFERENCES (1) Reed, R. E., DenBeste, M., and Humoiler, F. L., •. $oc. Cosmetic Chem., 1,109 (1948). (2) Brown, A. E., U.S. Patent No. 2,688,972. (3) DenBeste, M., and Reed, R. E., U.S. Patent No. 2,$40,980. (4) Hopkins, R., and Reed, R. E., U.S. Patent No. 2,682,272. (5) Kolb, J. J., and Toennies, G., .qnal. Chem., 24, 1165 (1952). THE BEHAVIOR OF PERFUME MATERIALS IN THIOGLYCOLATE HAIRWAVING PREPARATIONS* By EDWARD SAGARiN and MARVIN BALSAM Standard •lromatics, Inc., lYew York 3, N.Y. FEW COSMETIC preparations present problems so challenging to the perfumer as does the thioglycolate-based cold permanent wave. The sources of the difficulties facing the perfumer are numerous: 1. The strong odor of the thioglycolate salts, or of the impurities therein, requiring powerful odorants to give adequate coverage. 2. The strong odors that are liberated upon the reaction of the thio- glycolate salt with keratin and the subsequent rupture of the disulfide linkages in the hair. 3. The alkaline nature of the medium, excluding from use some organic materials generally used in perfumery, but unstable at a pH of 9 or over. 4. The reactive nature of thioglycolates resulting in the formation of reaction products with certain common constituents of perfumes and the powerful obnoxious odor, even in trace quantities, of some of these reaction products. 5. The relatively unstable nature of most thioglycolate emulsions, their tendency to separate or to cream, which is almost invariably aggravated by many perfume raw materials. * Presented at the May 18, 1956, Meeting, New York City.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)