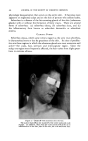
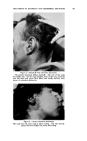
464 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS increase, in some cases markedly, the free sulfhydryl groups that may be detected in samples of dry, powdered plantar keratin. It was considered probable that the detergents disrupted certain cross linkages in the keratin molecule so as to expose previously enclosed --SH groups. The linkages so disrupted were not disulfide linkages because identical treatment of disulfide-containing materials such as cystine, insulin or keratin pretreated with iodoacetate yielded no--SH groups. Furthermore, lithium bromide, a hydrogen bond breaker, had the same effect as a detergent in increasing the --SH values of keratin (22). It is assumed that the--SH groups exposed by detergents were those cross linked by hydrogen bonds buried within the protein coils so tightly that they were unreactive. Flesch (10) has extended this line of investigation to include selenium sulfide and Selsun suspension. The data obtained are shown in the table below. INACTIVATION OF FREE SULFHYDKYL WITH SELENIUM SULFIDE-DETEKGENT SUSPENSIONS Inhibi- Incubation, tion, Tissue Preparation 40øC. % Mouse liver, 10 mg. Human epidermis, 10 mg. 0.5% SeS2 in Dupono], 0.5 mi. 1 hr. 80 10 min. 51 Selsun 5X diluted, 0.5 ml. 1 hr. 69 10 min. 44 0.5% SeS2 in Duponol, 0.5 ml. 10 min. 21 Selsun 5X diluted, 0.5 ml. 10 min. 5 It was concluded that the effect of Selsun suspension is twofold: The detergent first releases previously nonreactive sulfhydryl groups and the selenium compounds then inactivate some of these groups. These valuable data deserve to be studied and confirmed, if possible, by other investigations along parallel lines. Some experiments made in our own laboratories (23) have given in part results similar to those of Flesch, while others have given opposite results. For example, the block- ing of--SH groups in mouse liver homogenate by selenium sulfide was con- firmed. On the other hand, with a number of protein preparations in which --SH groups were inactivated by the usual sulfhydryl-binding re- agents, selenium sulfide showed no activity at all, or such low activity that it seemed to be merely a matter of total protein adsorption by the selenium sulfide. If it were conclusively proved that the selenium sulfide-detergent com- bination acts in the diseased scalp by binding sulfhydryl groups, such findings would obviously involve Selsun suspension with very fundamental body mechanisms. It is clear that the whole process of keratinization is greatly dependent on sulfhydryl-containing participants in the processes of
SELENIUM SULFIDE AS AN ANTIDANDRUFF AGENT 465 metabolism. Selenium compounds inhibit certain enzymatic reactions dependent on reversible sulfhydryl • disulfide changes also those sys- tems which require the presence of succinic dehydrogenase enzymes, con- taining free--SH groups. However, it must be remembered that this has been shown only for soluble compounds such as sodium selenite. Before attributing inhibition of those reactions to selenium sulfide, it must be shown that this highly insoluble material is absorbed by the skin in an active form. Finely divided sulfur is known to be directly absorbed by the skin and converted into S= and HS- ions (19, 24). It is quite possible that selenium sulfide is likewise absorbed in minute traces and converted into selenide and sulfide ions, the selenide ions then blocking the enzyme systems in- volved in the accelerated elaboration of immature epithelial tissue. We feel that this is a place where radioactive techniques are likely to advance the solution of the problem of mode of action. Exploratory studies that we have made support this view. With careful techniques one should be able to determine directly the extent to which selenium sul- fide tagged with radioactive selenium and/or sulfur is absorbed by the skin. The rate at which it leaves the site of application through systemic absorption might be correlated with the period of ef}ectiveness of the drug in inhibiting dandruff scale formation. Other lines of study will suggest themselves as the investigation proceeds. CONCLUSION Selenium sulfide-detergent suspension has proved itself of unquestioned value in a variety of seborrheic dermatoses, of which dandruff is an im- portant example. Data now available suggest several possible mecha- nisms by which the formulation may act, but more work must be done before the mechanism is elucidated. In the course of such investigations much light may be shed on the basic cause of dandruff itself. REFERENCES (1) Nordlander, B. W., U. S. Patent No. 1,711,742 and U.S. Patent No. 1,860,336. (2) Nordlander, B. W., U.S. Patent No. 1,860,320. (3) Baldwin, M. M., and Young, A. P., Jr., U.S. Patent No. 2,694,669. (4) Mattoon, R. W., Laboratory Records, Dept. of Physical Chemistry, Abbott Labora- tories. (5) Slinger, W. N., and Hubbard, D. M.,/lrch. Dermatol. andSyphilol., 64, 41 (1951). (6) Slepyan, A. H., Ibid., 65, 228 (1952). (7) Spoor, H. J., Drug & CosmeticInd., 77, 44 (1955). (8) Cohen, L. B., Arm. y. Ophthalmology, :38, 560 (1954). (9) Fritz, M. H., Clin. Med., 2, 695 (1955). (10) Flesch, P., y. Investigative Dermatol., 21,233 (1953). (11) Halter, K.,/lrch. Dermatol. and Syphilis, 178, 340 (1938). (12) Lemley, R. E., y. Lancet, 60, 528 (1940). (13) Spoor, H. J., Traub, E. F., and Bell, M., •lrch. Dermatol. and Syphilol., 69, 323 (1954). (14) Grundy, W. E., Laboratory Records, Dept. of Microbiology, Abbott Laboratories. (15) Shalt, H. M., Illinois Med. •., 105, 122 (1954). (16) Slinger, W. N.,Ibid., 105, 120 (1954).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)