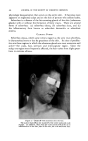
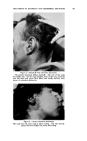
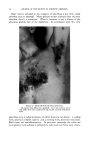
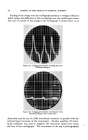
MEASUREMENT OF STATIC CHARGE ON HAIR 473 a week. These subjects were then reshampooed with the same shampoo followed by a creme rinse treatment and oscillograph measurements made daily for another week. Comparative results are shown in Table 2. In this particular study the antistatic effects were still quite visible on some heads on the fourth and fifth days after creme rinse treatment as may be seen by comparison with the results obtained with shampoo only. By the eighth day the effects had disappeared almost completely. According to Hayek (4) and to McLean (3) antistatic agents are pre- sumed to work through one of two mechanisms or possibly by a combina- tion of the two mechanisms: (a) by providing a film of inherently lower electrical resistance or (b) by coating the fiber with a material which attracts moisture from the air--an adsorbed water film having a lower resistance than the untreated fiber surface. Particularly if the second mechanism is the predominant one a marked effect of relative humidity on antistatic properties would be expected. Such data has recently been re- ported by Keggin (5) on the effect of relative humidity on static charge TASLE 3--ErrzcT or RELATIVE HUMIDITY ON STATIC CHAI•GE AFTER. USE Or SHAMPOOS OR. RISSE OS HATR. TR.ESS•S 27% R.H. 51% R.H. 76.5% R.H. Shampoo J Shampoo K Rinse C Rinse D Rinse E Rinse F 143 115 2O 26 25 38 10.8 3.3 6.8 2.2 0.8 0.4 1.3 0.4 1.5 0.4 2.1 0.5 using cotton and protein fibers. In Table 3 there is shown data on the effect of relative humidity on static charge development on hair tresses following treatment with several shampoos or rinses. As will be seen from the data and as might have been anticipated, static charge values are definitely lower at higher humidities both for the shampoos and for the rinses. At all three humidities, the effect of the cationic rinse is to cause a lower static charge than the shampoo alone. It is interesting to note that it is necessary to go to high humidities (about 75 per cent) to obtain low static charge values with shampoos similar to those obtained with cationics at much lower humidities. This may be due to the cationic treated hair adsorbing more water than untreated hair at any given humidity, thereby diminishing static charge. The question arises as to whether we are measuring an inherently lower static charge production or a more rapid rate of dissipation of the charge which is produced. This cannot be answered unequivocally at this time but from the data we have obtained, especially the low rate of charge leakage from charged tresses, we are led to believe that the primary effect of the cationics is to permit only a low charge build-up rather than to assist
474 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS in dissipation of the charge. The possibility that the effect of the treat- ment may also be that of reducing surface friction by deposition of a lubricating film should not be overlooked. We should also consider that the antistatic properties of cationics may be due not necessarily to greater effectiveness per se but also to the fact that they are quite substantive to hair and leave a greater deposit of material on the hair than other com- pounds which might be equally effective, weight for weight. It should not be inferred from the above discussion that the only effec- tive compositions for reducing static charge are the cationic creme rinses. Work has been done with other postshampoo treatments, some of which were found by the above techniques, to be quite effective in lowering static charge. In this category may be mentioned a commercial creme rinse product which, on analysis, was found to contain no cationic but to be based on a superratted anionic detergent. No attempt should be made from the data presented in Tables 1 and 2 to draw any indirect comparisons among the various shampoos as to their relative antistatic properties. These are studies of the effect of creme rinses and not a comparison study of the shampoos used. The actual peak values taken from the photographs will vary with the type of hair, with the length of hair, hair condition, relative humidity, method of comb- ing and probably many other variables so that the only valid comparisons which may be made among shampoos are such as were shown in Table 3 where shampoos were compared at the same time on swatches taken from a homogeneous tress (or compared directly in half-head tests). We have attempted, both by half-head technique on models and by direct comparison using purchased hair tresses, to compare shampoos for their relative antistatic properties. In general, it may be said that this program did not prove nearly as successful as the work on the effect of creme rinse. We have seen some differences in static charge development after different shampoo treatments. These differences have been on the whole, small and not statistically significant. Considerable difficulty has been encountered in reproducing results and frequently results would re- verse themselves or conflicting data would be obtained from the experi- ments on models and on tresses. We now believe that part of the difficulty in evaluating shampoos by this technique is inherent in the experimental set-up. It will be obvious that in the case of an essentially nonconducting substance such as hair or a comb only a portion of the total generated charge is picked up and trans- mitted to the oscillograph. While fluctuations in the per cent of the pick-up are probably of minor importance when comparing a good anti- static agent, such as a cationic, with an anionic shampoo these fluctuations may be of overriding importance when comparing two materials of more or less similar properties, such as two shampoos.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)