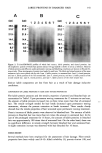
342 JOURNAL OF COSMETIC SCIENCE Table I Comparison of Amounts of Labile Proteins to Tensile Properties Treatments Labile proteins Tensile strength Extension at break (mg/g hair) (gram/hair fiber) (%) Untreated 0.251 _+ 0.016 115.8 _+ 26.8 45.6 + 6.8 Permed 15 min 0.604 _+ 0.115'* 83.7 -+ 25.1'** 53.1 + 9.9*** Permed 60 min 0.753 + 0.127'** 63.4 _+ 15.1'** 66.4 _+ 8.3*** Bleached 60 min 3.19 _+ 0.49*** 104.1 + 28.8 Ns 47.5 _+ 7.2 Ns Bleached 16 hr 51.7 _+ 4.6*** 106.1 _+ 25.1 Ns 51.8 _+ 7.7** All values denote the mean _+ SD. Four experiments were performed for labile proteins. Forty fibers were subjected to the measurement of tensile properties. Statistical analyses were performed by t-test to the values of untreated hair. NSp 0.05. **p 0.01. ***p 0.001. the abrasion method (11-13) are all based on measurement of protein loss. In this study, we have shown that the lablie protein levels in hair drastically increased upon permanent waving or bleaching treatments. The amount of lablie protein is a useful index for hair damage. This index has several superior characteristics compared to other indexes men- tioned above. Protein elution, which previously was reported to be correlated with damage to hair (10), was compared with the labile protein. The amounts of eluted proteins increased to a lesser extent than those of the lablie proteins under conditions of permanent waving treatment. Furthermore, the labile protein levels also drastically increased upon bleach- ing treatment, whereas the protein levels eluted from bleached hair were very low. The alkali solubility test is a classical method for assessing hair damage. Hair damage has been reported to correlate with the amount of hair proteins extracted with alkaline solution (e.g., 0.1 N sodium hydroxide). The protein loss estimated through the mea- surement of the extracted hair sample weight was 7.6% of total untreated hair, whereas the loss from the bleached hair was 12.4% (9). This blunt response, compared to the wide dynamic response of labile proteins (Table I), seems to be insufficient to accomplish reproducible assessment of hair damage. Damaged hair has been considered more susceptible to abrasion than intact hair. Quan- titative measurement of the protein lost during hair sample shaking in water (11) or abrading using a comb (12,13) was reported to be a simple and sensitive method for assessing surface damage to hair. However, there might be some problem with the abrasion method due to the technical difficulty in reproducing the shaking conditions or the hair brushing conditions. Tensile properties have been widely used for the estimation of hair damage. Consistent with the previous report (6), our result, measured in a dry condition (at 60% relative humidity), showed that the tensile property changes were clearly induced in permanent waved hair. On the other hand, the tensile property fluctuated only slightly in bleached hair. It had been reported that tensile property changes from bleaching were more clearly detected in wet conditions (8). Even though tensile properties are affected to some extent by dry or wet measurement conditions, tensile property changes by bleaching tend to be
LABILE PROTEINS IN DAMAGED HAIR 343 small compared to those induced by perming (6,7). As the labile protein levels changed drastically upon bleaching treatments, this index is superior, especially for damage assessment of bleached hair. Hair releases larger amounts of protein under reducing conditions than under oxidative conditions. Permanent waving treatment is performed to realign the cross-linked disul- fide bridges of hair proteins by reduction, whereas bleaching lotion is designed to decolorize melanin pigment by oxidation. Consequently, the tensile property of permed hair is worse than that of bleached hair (6,7). On the other hand, our results show that labile proteins accumulate in hair upon bleaching treatment as well as permanent waving treatment. Considering that no significant difference was observed in the elec- trophoretic patterns of labile and eluted proteins, a part of labile hair proteins is likely to elute from hair. A hypothetical process for the formation, accumulation, and release of labile protein is delineated in Figure 3. We propose that the molecular or micro- environmental modification of hair proteins (i.e., conversion from stable to labile state) upon permanent waving and bleaching treatments is an important process leading to serious hair damage. The rate of conversion to labile proteins is larger than the release into the effluent. Bleaching treatment especially releases a very small amount of hair protein into the effluent. As a consequence, the repetitive permanent waving or bleach- ing treatment accumulates the labile protein in internal hair. The molecular conversion from stable state to labile state corresponds to the initial step of the hair damaging process. Therefore, it may be possible to assess hair damage from an early stage through the increase of lablie protein in hair. We previously characterized hair protein components eluted under reducing and oxi- dating conditions. S100A3, a soluble cuticular protein, was found in the permanent waving lotion (14). A major component in the permanent waving lotions and bleaching agents was identified to be ubiquitin, which is ubiquitously distributed in the hair follicle (4). Both proteins occupy a large percentage of the labile protein fraction (3), but decrease from the tip side of hair, probably resulting from loss during daily hair treat- ments (4,14). In this study, we show drastic changes of labile proteins upon chemical Hair Perm rm I•• kabi•: =I: Elute• Bleach Bltach Figure 3. Hypothetical process of molecular or environmental modification of soluble proteins in perreed and bleached hair. Upon permanent waving or bleaching treatments, soluble proteins in the stable state are transformed into the labile state. However, almost soluble proteins are retained in internal hair, because the amount of the protein conversion is much larger than the protein elution. Resultant labile proteins can be isolated under the partial extraction (mild reducing) condition.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)