58 JOURNAL OF COSMETIC SCIENCE candidate because it was revealed that paeoniflorin, in our preliminary experiments, suppressed the expression of senescence-associated 13-galactosidase. Peony root (Paeoniae radix) is one of the most well-known herbs in China, Japan, and Korea, and has long been used as a component of traditional medicinal prescriptions. Several compounds including oxypaeoniflorin, albiflorin, benzoylpaeoniflorin, and pae- oniflorin are reported to be contained in peony root (6). Among them, paeoniflorin is known to be one of the principal bioactive components of peony root and has been reported to have immunoregulatory (7), anti-inflammatory, anti-coagulant (8), and seda- tive (9) effects. Recently, we developed a new cosmetic ingredient, partially purified paeoniflorin (PF), from the roots of P aeoniae lactiflora. It could be characterized by enriched paeoniflorin content (more than 60%), and Figure 1 shows the molecular structure of paeoniflorin. To verify the potential usefulness of PF as a cosmetic active ingredient, we investigated the effect of PF on the DNA damage caused by UVB irradiation. We also performed a clinical study with a cosmetic formulation containing 0.5% PF. In this report, we present the protective effects of PF on DVB-induced DNA damage as well as its anti-wrinkle activity in vivo. EXPERIMENT AL PREPARATION OF PARTIALLY PURIFIED PAEONIFLORIN (PF) PF was extracted and partially purified from the roots of Paeonia lactiflora. Briefly, peony root was ground and extracted with 7 5 % ethanol. The extract was concentrated by evaporation and resuspended in distilled water and fractionated with ethyl ether. The water-soluble part was loaded into a silica gel column, and the fraction containing peoniflorin was resolved with chloroform acetone (4: 1) as moving phase. The purity of paeoniflorin was determined to be about 64% by HPLC. CELL CULTURE Normal human keratinocytes (NHK) were cultured from adult foreskin. NHK cells were maintained in keratinocyte growth medium (KGM, Clonetics, USA) in a humidi- Figure 1. Molecular structure of paeoniflorin (C23H28O 11 M.W. 480.45), major constituent of PF.
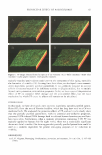
PROTECTIVE EFFECT OF P AEONIFLORIN 59 fied incubator at 3 7°C under 5% CO2. Cells less than fifth passage were used for this study. CELL TREATMENT AND UV IRRADIATION NHK cells were cultured for 12 h in KGM containing various concentrations of pae- oniflorin. Then the medium was replaced with phosphate-buffered saline (PBS) without calcium and magnesium, and the cells were irradiated with 60 mJ/cm2 of UVB (peak emission 312 nm) using the Biotronin UV irradiation system (Vilber Lourmat, France). The cells were then harvested and subjected to a comet assay. ANIMAL EXPERIMENT Eight-week-old male hairless mice weighing 27-33 g (DaeHan Biolink, Taejeon, Korea) were used in this study. The animals were divided into five groups, with two mice in each group, and the dorsal skin was treated topically with various concentrations of paeoniflorin dissolved in 80% ethanol. The samples were applied twice a day for six days. After the last application, the hairless mice were anaesthetized by an intraperitoneal injection of sodium pentobarbital (50 mg/kg body weight) and remained anaesthetized until the mice were sacrificed. The mice were exposed to sham or 1 J/cm2 of UVB. After the mice were sacrificed, the skins were excised and rinsed with Hanks Balanced Salt Solution without calcium and magnesium (HBSS, Gibco BRL, USA). The keratinocytes were isolated by the method ofYendle et al. (10), with some modifications, and the cells were subjected to a comet assay. COMET ASSAY The comet assay for detecting DNA damage was performed according to the procedure of Singh (11). The slides for the comet assay were observed at 250 x magnification in a Metallux-3 fluorescence microscope (Leitz, Germany) with a color CCTV camera (Panasonic, Japan). Fifty images were randomly selected from each sample, and comet tail moment was measured according to the procedure of Konca et al. (12). The comet tail moment is positively correlated with the level of DNA breakage. The mean value of the tail moment for a particular sample was taken as an index of DNA damage. CLINICAL TRIAL A conventional cosmetic preparation containing 0.5% PF was used in the clinical study. Twenty healthy female volunteers aged 30 to 56 (mean 42.5 years) participated, with informed consent. All of them had more than a moderate degree of fine wrinkles around their eyes. The study was performed in a randomized, placebo-controlled double-blind trial. After randomization, half the face was treated twice daily for eight weeks with PF cream and the other half with the control cream (placebo). The clinical efficacy was evaluated by low-coherence interferometry, described elsewhere (13). Briefly, skin rep- licas of the eye rim region (crow's feet) were made at the start of the study and after four weeks and eight weeks of sample application. The replicas were scanned for three- dimensional profiles and analyzed with Microfocus (UBM, Germany). The parameter for
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)