80 JOURNAL OF COSMETIC SCIENCE A COMBINATORIAL INVESTIGATION OF THE EFFECTS OF ORDER OF ADDITION IN THE INERACTION OF POLYELECTROLYTES WITH SURFACTANTS Robert Y. Lochhead, Ph.D., Lisa R. Huisinga, Christina Edwards and Anthony Hill The Institute for Formulation Science, School of Polymers and High Performance Materials, The University of Southern Mississippi, Hattiesburg, MS 39406-0076 INTRODUCTION Formulators of complex mixtures have long known that the characteristics of their final fonnulation and the position of "equilibrium" often depends critically upon the order of addition of ingredients and the precise processing conditions under which the fonnulation was made. The large variety of possible outcomes derive from the many eigenstates that are available to each composition of a complex mixture due to the fact that the bonds between the component molecules are weak physical bonds and therefore a potential multitude of nanostructures can be fonned. This is especially true of systems comprising polyelectrolytes and oppositely charged surfactants in the semi-dilute regime, because both polyelectrolyte conformation and surfactant micellar association structures are strongly influenced by the ionic environment of the polymer and surfactant molecules. This is especially important for 2-in-l shampoos that depend upon the spontaneous creation of a polyelectrolyte/surfactant coacervate to deposit active conditioning, styling or antidandruff ingredients during the shampoo process. The investigation of the effects of order of addition requires exanimation of a myriad of samples and it is virtually impossible by conventional techniques. This task is ideally suited to investigation by a combinatorial approach aimed at the generation of libraries of pseudo-phase diagrams. In this study we developed a high-throughput screening method to generate phase diagrams over a large range of concentrations for cationic polysaccharide interaction with anionic surfactant in the presence and absence of dissolved electrolyte. Using a liquid handling system for sample preparation, we are able to analyze nearly 1000 samples per day, making the above goals of understanding electrolyte effects and coacervate structure-property relationships attainable. EXPERIMENT AL Materials. Commercially available Polyquatemium-10 polymers of varying molecular weights and charge densities were obtained from Amerchol and used as received. The anionic surfactant sodium lauryl ether (3EO) sulfate, 28.82% actives, was obtained from Stepan Company. Sodium chloride (NaCl), A.C.S. certified grade, was used as received from Fisher Scientific. Distilled, deionized water was used in all samples. Sample Preparation. The liquid handler used for high-throughput fonnulation has a viscosity limitation of lOOOcps and cannot distribute solids. To overcome these constraints, pre-mixes of the polymer and surfactant were made. Polymer pre-mixes were made by slowly adding polymer powder to water under constant agitation and low heat. These pre-mixes were preserved by 0.003% actives Kathon CG. Surfactant was supplied as a viscous liquid and was diluted with water to make pre-mixes of a given % actives. Pre-mixes of sodium chloride were made by dissolving NaCl solids in water. Formulation samples were prepared using a Beckman Coulter Biomex FX Laboratory Automation Workstation. Programs were designed to create over 350 samples in less than one hour, with repetitions included to ensure accuracy. 96-well plates with glass vials are used for sample vessels. Once all materials are delivered to the 96-well plates, the samples are mixed using a 96-well plate attachment on a Scientific Industries Vortex Genie 2. Phase Separation Analysis. Phase separation is used to indicate the relative amount of coacervate fonned in each sample. UV-Vis absorbance measurements are taken on each well in the 96-well plate using a Tecan Satire Multifunction Multiplate Reader. When a new system is first subjected to this analysis, an absorbance scan from 230-990nm (20nm step) is performed on wells that show no, little and high amounts of visible phase separation in order to determine the most desirable fixed wavelength for absorbance readings. For cationic polymer systems, the desired wavelength is 410nm. Absorbance readings are
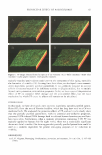
2005 ANNUAL SCIENTIFIC MEETING performed on all wells of the 96-well plate, with an average total analysis time of less than 2 minutes. The absorbance units are reported and plotted as a function of% actives polymer and% actives surfactant. Coacervate Structural Analysis: Association colloids can exist in a variety of structures, including liquid crystalline-like structures. Micellar and cubic phases are isotropic and do not exhibit birefringence, whereas lamellar and hexagonal phases are anisotropic and do exhibit birefringence. To rapidly analyze samples in 96-well plates for possible structure, a pass/fail birefringence test was developed. The samples are placed on a light box between crossed polarizing sheets. Those samples that do rotate light (birefringent) show light through the polarizing sheets and samples that do not rotate light are black through the polarizing sheets. The pass/fail results of this test are reported as a function of% actives polymer and% actives surfactant. More detailed analysis of the samples that exhibited birefringence was performed using Polarized Light Microscopy (PLM). In PLM, the sample is placed between crossed polarizers and the magnification allows for the definition of different structures. Lamellar and hexagonal liquid crystal phases were identified by the characteristic focal conic images that are representative of each structure. Visualization of Data: High throughput screening generates an enormous amount of data in a very short time. Analysis Of results by conventional methods would slow the pace of the investigation. Therefore, it was necessary to devise techniques for quickly visualizing large arrays of data. We chose to do this by color-coding our measured parameters and by presenting the data in the form of a color-coded composition map. RES UL TS AND DISCUSSION Polymer-Surfactant Interactions. Initial high-throughput studies were performed on three component systems cationic polymer, anionic surfactant and water. This established a baseline for further studies (e.g. electrolyte effects) and also can be used as a predictive model coacervate amount and structure for cellulosic pol ym ers of varying charge densities and molecular weights. The properties of the cellulosic polymers studied are shown in Table 1. T l IC . abe attomc ce ll I u os1c po1vmer properties POLYMER MOLECULAR CHARGE WEIGHT DENSITY UCARE LK 350,000 0.357 UCARE 350,000 0.678 LR400 UCAREJR125 350,000 1.321 UCAREJR400 450,000 1.321 UCARE 1,800,000 0.678 LR30M UCARE 2,000,000 1.321 JR30M The coacervate amount varies significantly as a function of both charge density and molecular weight. In the Phase Separation contour phase diagrams, the amount of coacervate is indicated by the color scale, where blue represents no coacervate and red means high amounts of coacervate. From the results in Figure 1, low molecular weight polymers produce low amounts of coacervate regardless of charge density. At a slightly higher molecular weight and high charge density, coacervate formation is increased as evidenced by the comparison of the JR400 diagram to the JR125 diagram. At high molecular weights, LR30M and JR30M, coacervate amount is higher than low molecular weight systems with the same charge density, LR400 and JR125, respectively. In addition to molecular weight effects, at higher molecular weights an increase in charge density also increases the amount of coacervate produced. By mapping the phase separation results as done in Figure 1, we are able to predict the amount of coacervate if given a molecular 81
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)