256 JOURNAL OF COSMETIC SCIENCE Abil EM 90, W/O an emulsifier. Cetyl dimethicone copolyol. Supplied by Th Gold- schmidt Ag (Essen, Germany) Abil WE 09, a W/O emulsifier. Cetyl dimethicone copolyol (and) polyglycerol-4- iso-stearate (and) hexyl laurate. Supplied by Th Goldschmidt Ag (Essen, Germany) Abil B8845, polysiloxane polyether copolymers, a silicone surfactant with a watery external phase (O/W). Supplied by Th Goldschmidt Ag (Essen, Germany) LSNa, a surfactant with a watery external phase (O/W) n-decane Distilled water METHOD Several formulations were elaborated based on the formula of Vanderhoff et al. (10) and modifying the surfactant (Table I). The following formulations were chosen: LSNa (O/W anionic surfactant), Abil 8851 (W/O silicone surfactant), Abil EM 90 (W/O silicone surfactant), and Abil WE 09 (W/O silicone surfactant) at different concentrations. We obtained formulations that were studied for both organoleptic properties and sta- bility, verifying that the formulas with Abil EM 90 and Abil WE 09 provided the best characteristics, and in these formulations we studied the influence of the concentration of emulsifier (Table II). Test release. Ointments containing Aloe vera were placed in a 6-cm petri dish with lightly acidic water and shaken at 60 rpm at 37øC in a thermosrated bath. This shaking speed was shown to be sufficient to mix the Aloe vera throughout the aqueous phase after its release from the vehicle, without leading to significant dissolution of the base. The amount of Aloe vera released was determined after 0.5, 1, 2, 4, 6, 8, and 24 h by measuring absorption at 256 nm with a Perkin-Elmer running lambda 2 mod.127 spectrophotometer. Blanks containing no active principle were prepared in the same way as samples, and tested to compensate for possible interference between the components of the formulation. At least three determinations were done for each data point. RESULTS AND DISCUSSION The pH was measured with a direct method using a Crison mcropH 2001 meter (Alella, Barcelona, Spain) (11). In addition, we used phase separation with n-butanol and tri- Table I Formulations Modifying the Surfactant Formulation 1 Formulation 2 Formulation 3 Formulation 4 DC 245 24.6% 24.6% 24.6% n-decane 0.37% 0.37% 0.37% H20 73.98% 73.98% 73.98% LSNa + 1% -- -- Abil B 8851 -- 1% -- Abil EM 90 -- -- 1% Abil WE 09 -- -- 24.6% O.37% 73.98%
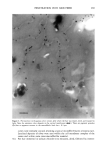
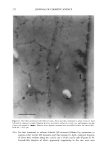
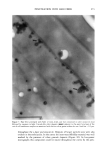
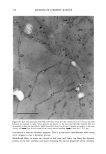
SILICONE VEHICLES FOR ALOE VERA 257 Table II The Influence of the Concentration of Emulsifier Formulation 3 Formulation 4 Formulation 5 Formulation 6 DC 245 n-decane H20 Abil EM 9O Abil WE O9 Abil EM 90 Abil WE O9 24.6% 24.6% 23.3% 23.3% 0.37% 0.37% 0.3% 0.3% 73.98% 73.98% 70.98% 70.98% 1% -- -- 1% 5% -- -- 5% chloromethane (12). The pH of the formulas selected was determined and found to be constant over time at a value of pH 5.5 + 0.5. MICROPHOTOGRAPHIC STUDY It is well known that the smaller the droplet size, the greater the stability of the system. Therefore, to study stability as a function of time and temperature, the formulations were characterized by determining the droplet size via light microscopy (Olympus BX 40 microscope coupled with an Olympus SC 35 camera). Figures la and lb are photographs obtained from preparations with Abil EM 90 sur- factant at 1% and 5% and Figures 2a and 2b are photographs of the Abil WE 09 surfactant at 1% and 5%. A 5% concentration was found to result in small droplets, regardless of the surfactant, although the photos of Abil EM 90 reveal greater homo- geneity and even smaller droplet size. Moreover, the study of the influence of time and temperature has revealed Abil EM 90 to provide slightly more stable preparations. Figure 3 shows the distribution of the droplet size for both Abil EM 90 and Abil WE 09, clearly revealing the above-mentioned characteristic of a smaller droplet size at greater concentrations of surfactant. In Abil EM 9, 80% of droplets range in size from 1-9 pm, compared to 75% for Abil WE 09. When the surfactant concentration is lower, the increase in droplet size is considerable, as can be seen. Abil EM 90 1% Abil EM 90 5% (b) Figure 1. Microphotographs of the droplets of the formulations with Abil EM 90 at 1% (a) and 5% (b)
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)