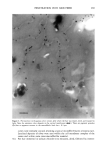
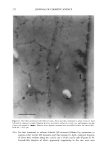
352 JOURNAL OF COSMETIC SCIENCE the release of odorant ingredients by measuring their diffusion rate to the air. In other cases, the solubility of the system in the skin is more important than the diffusion of the odorant to air. Therefore the analytical method is based on the specific need. Preparation of NanoSal TM NanoSal TM consists of a solid core, composed of a blend of hydrophobic crystalline polymers, and a high molecular weight polymeric surfactant with a charge group that is embedded on the surface of the sphere and assist in adhering the NanoSal TM to biological surfaces such as: skin, and hair:. The polymers used for the matrix are biodegradable, and made out of materials that are listed as GRAS for cosmetic application. The polymeric surfactants provide the solid core comprising the active ingredients, the bio-adhesive properties. The NanoSal TM particles diameter is between 0.01 microns and 5 microns. Particle size distribution is controlled by type of polymers, and production conditions. The NanoSal TM size and hydrophobic properties closely related to those of liposomes, and thus are used to enhance lipophilicity of actives and penetration into the deeper layers of the stratum cornium. The major benefit of the NanoSal TM over typical liposomes is stability and ability to retain materials in its semi crystalline structure that is more resistant to shear, environmental changes and temperature. Other unique properties reside from the polymeric surfactants surrounding the surfaces of the nanospheres and include: 1, enhanced retention of the active in the core, through adding another layer that retains actives from leaking out the solid core during storage. 2, the lingering polymeric chain is used as a mean to enhance adhesion onto different surfaces, and 3, prevent the spheres from coagulation. The ability of the NanoSal TM to adhere onto human cells was studied utilizing HeLa cultured cells. The adhesion of the NanoSal TM onto the cultured cells is demonstrated in Figure 1. The images indicate that the NanoSal TM adhere specifically to the HeLa cells and less to the protein coated plastic surface. Figure 1: Specific adhesion of NanoSal TM to HeLa cells. Performance of NanoSal TM in practical application The analysis and visualization of ]NTanoSal TM adhesion and disintegration into a skin tissue is under process. We focused on demonstrating the adhesion of the system onto hair from typical hair care products (i.e., shampoo and conditioners). NanoSal TM slurry was easily incorporated into a typical commercial shampoo and conditioner products, resulting with a compatible and stable blend. Hair was used as a model, because the hair surface has some properties closely related to skin, and SEM can be utilized to determine the adhesion onto the hair. Hair swatches
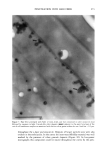
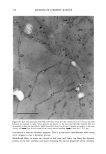
2001 ANNUAL SCIENTIFIC SEMINAR 353 were washed with shampoo comprising the NanoSal TM, and analyzed by SEM. Figure 2 shows the adhesion of NanoSal TM onto hair from a shampoo application. Figure 2: Adhesion of NanoSal TM to hair from shampoo application. Conclusions A rational scientific approach is key to develop effective controlled release systems for skin care products. The process of developing new controlled release systems for skin has several major steps: screening and selection of materials, processing optimization and evaluation of performance. ACRS based on bioadhesive nanospheres was designed to deliver a wide range of active ingredients. The key to success is tailoring the system to the specific application. References 1. S. Shefer et. al., Controlled Release Society, Annual meeting in Paris 1999 2. Patent application, 2000 to Salvona LLC 3. Crank J. and Park G. S., Diffusion in Polymers, Academic Press, New-York, 1969 4. Konos W., Ed. Barrier Polymers and Structures, ACS Series, Washington DC, 1990
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)