350 JOURNAL OF COSMETIC SCIENCE CONTROLLED RELEASE SYSTEMS FOR SKIN CARE APPLICATIONS Sam Shefer, Ph.D. and Adi Shefer, Ph.D. Salvona, LLC- Controlled Release Technologies • Abstract The major challenge in developing effective personal care and cosmetic products, on the physiological level, is the exposure of receptive site to effective levels of ingredients over a long period of time. Several studies have shown that active ingredients, commonly used for cosmecutical applications, including: vitamins, co-enzymes, enzymes, amino acids, and polypeptide, are more effective if they are available at effective level at the target site. This challenge can be addressed by either repetitive applications, or alternatively by utilizing advanced controlled release systems (CRS). CRS are designed to sustain the release, and target the active ingredients onto a specific site. Pharmaceutical CRS have significantly advanced drug efficiency and generated many successful commercial products. The knowledge and experience gained in the pharmaceutical field allows us to better understand systems, and design cost effective CRS to meet cosmetic and personal care products requirements. The system reported in this paper is based on sub-micron nanospheres specially design for skin care. In addition to its beauty and tactile properties the nanosphere-based product provides an efficient carder system for cosmeceutical actives to reach the deeper layer of the stratum cornium through the small size of the nanospheres and their hydrophobic properties that allow them to dissolve within the tissue and release their content. The results reported in this paper provide an overview of the rational in designing the product. Introduction Several research studies have demonstrated that the effect of specific active ingredients, such as: cosmecutical, on skin depends on the length of time the active is exposed to the tissue, at an effective level. The major challenges in enhancing the performance of various actives in skin care products are safety, stability, and efficacy. Advanced controlled release technologies may be a practical approach to address these issues. Common encapsulation technologies enable to retain active ingredients and maintain their integrity and stability over a long period of time. The system discussed in this paper was designed to provide a triggered release (release is activated by the consumer or through environmental change) and targeted release (release is targeted to specific action site). In addition to its delivery properties, the system retains the active in its natural form over extended period of time in the product base under accelerated stability tests. The objective of this paper is to discuss the rational approach to address the fundamental requirements in designing advanced controlled release systems (CRS) for skin care. This paper focuses on the development of nano-sphere technology with unique properties and performance features for skin care. Methods and Materials Salvona's proprietary nanospheres, NanoSal TM, are produced 2 by liquidation of a blend of hydrophobic polymers with the active to be released. The blend is stirred under high speed and high pressure and injected into a reactor containing a water-based emulsion of a blend of diblock polymers (both long molecular hydrophobic and hydrophilic chains). The slurry is transferred through a heat exchanger to reach a temperature of -5 ©C and lower for a short period of time. The suspension is allowed to reach equilibrium at room temperature. The resulting product of this process is a stable suspension. All components of the NanoSal TM including the blend of polymers and the emulsifying polymers are tailor made per properties of the active, and chosen to meet the final performance requirements of the product. • Salvona LLC - Controlled Release and Advanced Technologies, 65 Stults Rd, Dayton, NJ 08810-1523, Tel: 609- 655 0173, Fax: 609- 655 9291, Email: Sam@Salvona.com
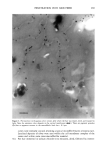
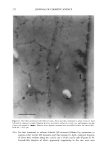
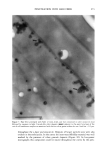
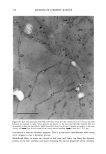
2001 ANNUAL SCIENTIFIC SEMINAR 351 To test the adhesion properties of the NanoSal TM to human cells, we used an ex-vivo method. The HeLa cell line, that we have been chosen to determine the bioadhesive properties of the NanoSal m' is an epithelial-like cell line, originally derived from a carcinoma of the cervix. To test the adherence of NanoSal TM to the cell surface, HeLa cells were plated at a density of 2x l0 s cells per dish (35 mm) in 2 ml medium. On the following day, the NanoSal TM were dispersed in 1 ml medium. The medium in which the cells were cultured were aspirated and replaced immediately with the NanoSal TM containing medium. The NanoSal TM were left to adhere to the cells by gravity for 5 min. At each time point the medium was aspirated, and the cells surface was gently rinsed twice with 2 ml medium. Evaluation of the performance of NanoSal TM from a hair shampoo was carried out by suspending the NanoSal TM slurry in a commercial shampoo base to a concentration of 5 ml/L. 5 gr of the shampoo comprising the NanoSal TM was applied to 5-gr hair swatches and rinsed well for 3 min with tap water to simulate typical application. The hair was allowed to line dry at room temperature for 48 hr prior to subjecting them to the Scanning Electron Microscope (SEM) for visual analysis. Results and Discussion The rational design of a controlled release system for skin care was directed to address several basic requirements from the aesthetic, practical and functional points of view. These issues include means to provide: good skin fell, conventional manner of application, performance that consumer can perceive and relate to, and cost effective product. We identified several major steps in the developmental process, including: 1. Mass transfer analysis to enable us to screen and select materials that retain the active during storage, and release it when it's in contact with skin. We wanted the system to be soft enough to allow the user to activate the release by pressure formed while massaging the cream onto the skin. 2. Materials that are safe, preferably selected from natural sources, and can meet the physical requirements for the process to manufacture NanoSal TM 3. Materials that retain the natural biological potency of the actives over time, stabilized active ingredients in aqueous cosmetic dermatological, and pharmaceutical compositions, over an extended period of time 4. System that can be incorporated in a typical commercial product base to result with a stable homogeneous product over time, as determined by accelerated standard stability tests 5. System adheres to skin or hair, from an application, to allow the release of the active over time following the application 6. Enhanced liposolubility of hydrophilic actives, thus facilitate uptake into cells and increase bio-availability and intracellular accumulation 7. Controlled, continuous release of effective levels of the active over an extended period of time following application 8. The release rate of the active ingredient can be synchronized with that of a "sensory marker" to convey to the consumer the product performance This paper reviews only some of the results obtained through this research, designed to develop a general prototype system. The research utilized specific actives to demonstrate the principles of the design. These principles can be applied for the design of various systems, to release of a wide range of botanical extracts, such as: peptide, vitamins etc. However, the properties of the active will determine the type of materials and procedures used for the preparation of NanoSal TM. We characterized the mass transfer coefficient of a model active through several blends of polymers that were screened as potential matrices for our NanoSal TM system. As an experimental model for a cosmeceutical active ingredient we used a blend of botanical extracts. This blend can also be used as a "sensory marker", due to the distinct odor of the materials. The sensory marker is used to simulate and convey the release of the active to the consumer. For a specific study we utilized a blend of 3 botanicals that have distinct odor, such as: lavender, orange extract and bergamot. The odorant release rate can be measured by analytical methods, such as: GC, HPLC, weight loss, as well as by sensory methods, by evaluating odor intensity. In this experiments, we evaluated
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)