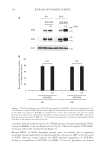
194 JOURNAL OF COSMETIC SCIENCE accordance with our previous report (13), UVA/B exposure decreased intracellular NAD+ content in NHEKs at the irradiation level of 19 J/cm2 UVA and 35 mJ/cm2 UVB, NAD+ levels were below 20% of control levels (Figure 3). Because SIRT1, an NAD+-dependent enzyme, plays an essential role in regulating autophagic protein degradation, we hypothesized that a decrease in SIRT1 activity because of NAD+ deficiency would suppress the SIRT1-mediated degradation of AGE-BSA, leading to accumulation of AGE-BSA in NHEKs. To test this hypothesis, we assessed - AGE-BSA UV AGE UV - AGE-BSA h 8 h 24 Actin - - - - 95 72 43 A B BSA 95 72 24h 8h 0 20 40 60 80 100 - - UV - - UV AGE-BSA AGE-BSA N.S. (kDa) - - N.S. Figure 1. UVA/B irradiation promotes AGE-BSA accumulation in NHEKs. Cells were irradiated with 13 J/ cm2 UVA and 23 mJ/cm2 UVB in phenol red-free EpiLife® medium and then cultured in an EpiLife® medium in the absence or presence of AGE-BSA (1.5 mg/ml) for 8 or 24 h. After incubation, the cells and culture medium were subjected to immunoblotting analyses and measurement of residual AGE-BSA, respectively. Data are expressed as means ± SEMs (n = 3) the means are expressed as a percentage of maximum residual AGE-BSA in the medium not taken up by cells. Residual AGE-BSA in the medium (% of maximum residue)
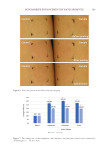
195 Accumulation of Advanced Glycation End Products whether the absence of SIRT1 activity affects intracellular AGE deposition. As shown in Figure 4A, treatment with a SIRT1 inhibitor, EX527, significantly increased intracellular AGE-BSA content in a dose-dependent manner. In contrast, SIRT1 activators, SRT1720 and resveratrol, decreased the level of subcellular AGE-BSA (Figure 4B). In addition, a decrease in AGE-BSA deposition was also observed when using an NAD+-boosting agent, NMN. These results suggest that SIRT1 plays a key role in controlling intracellular AGE accumulation. We were interested in whether SIRT1 affects the expression of the AGE-BSA CQ Baf AGE Actin 95 72 - - 43 (kDa) Figure 2. Exogenous AGEs incorporated into NHEKs are degraded via autophagy. NHEKs were preliminarily incubated in the EpiLife® medium in the presence or absence of an agent (1 µM of bafilomycin A1 or chloroquine) and then cultured in the absence or presence of AGE-BSA (1.5 mg/ml) for 8 or 24 h. After incubation, the cells were subjected to immunoblotting analyses. Abbreviation: CQ, chloroquine and Baf, bafilomycin A1. 0 20 40 60 80 100 120 0 UVA + 0 UVB 6 UVA + 12 UVB 13 UVA + 23 UVB 19 UVA + 35 UVB UVA (J/cm2) and UVB (mJ/cm2) doses ** ** ** Figure 3. Cellular NAD+ level in NHEKs rapidly decreases after UVA/B irradiation. NHEKs were exposed to UVA/B irradiation at the indicated dosages. The cellular NAD+ levels were measured 1 h postirradiation using NAD+/NADH assays. Data are expressed as means ± SEMs (n = 3) the means are expressed as numbers of fold-change compared to that of control cells. **p 0.01, in comparison to the control. Relative NAD+levels (%)
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)