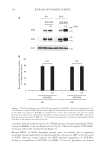
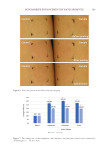
198 JOURNAL OF COSMETIC SCIENCE We also showed that UVA/B irradiation affects the mRNA expression of the AGE- detoxifying enzymes, GLO1 and GLO2 (Figure 5). GLO1 gene expression is transcriptionally regulated by nuclear factor erythroid 2-related factor 2 (Nrf2) (17). Nrf2 has been reported to be positively related to SIRT1 activity (24). UV irradiation induces NAD+ depletion through poly adenosine diphosphate-ribose polymerase activation (13), suggesting a temporal decrease in SIRT1 activity. Thus, impairment of the SIRT1-Nrf2 axis by UV exposure seems to decrease GLO1 gene expression in keratinocytes. As expected, the SIRT1 inhibitor, EX527, inhibited the mRNA expression of GLO1 in NHEKs (Figure 4). Moreover, using EX527, we have also shown that GLO2 expression negatively correlates with SIRT1 activity (Figure 4). It is well known that various stresses such as UV exposure increase the expression of defensive proteins against stress-induced damage (25). GLOs are responsible for detoxifying precursors of AGEs such as methylglyoxal (26). Radjei et al. have reported that GLO2 expression in the epidermis negatively correlates with AGE deposition (17). Considering these facts, UVA/B irradiation seems to induce the expression of GLO2 as a defensive response. In contrast, long-term UV exposure has been reported to critically reduce GLO2 protein levels in the epidermis (17). To examine how chronic UV irradiation affects GLO2 expression at the cellular level, NHEKs were exposed to UVA/B for 5 consecutive days. However, we did not find any difference in GLO2 expression between UV-exposed and unexposed cells (data not shown). Radjei et al.’s report has further shown that GLO2 expression remains unaltered in sun-exposed and sun-protected skin areas in young donors (17). Skin photo-aging gradually occurs over years through multiple and complex processes such as the loss of stem cell diversity (27). Thus, GLO2 decline in the epidermis is likely to appear only after years of UV exposure. In addition, as resveratrol and SRT1720 are considered to be anti-aging components that activate SIRT1, future studies that examine how these components can affect the decrease in GLO2 expression during such long-term UV exposure and may help clarify the mechanism of action of skin aging. In conclusion, this study revealed that SIRT1 reduces intracellular AGE content in keratinocytes. In addition, we have shown that SIRT1 activity is positively and negatively associated with the expression of GLO1 and GLO2, respectively. UV irradiation is known to affect SIRT1 activity and expression in the skin (13,28). Therefore, maintaining SIRT1 levels in the epidermis is a plausible approach for preventing photo-aging. ACKNOWLEDGMENTS We would like to thank Editage (www.editage.com) for English-language editing. AUTHOR CONTRIBUTIONS Conceptualization: KT-N. Validation: TK, KT-N. Investigation: TK, NK, TN, KT-N. Methodology: TK, KT-N. Writing (Original Draft): KT-N. Writing (Review & Editing): TK, NK, TN, KT-N. Visualization: TK, KT-N.
199 Accumulation of Advanced Glycation End Products Supervision: KT-N. Project administration: KT-N. REFERENCES (1) R. Singh, A. Barden, T. Mori, and L. Beilin, Advanced glycation end-products: A review, Diabetologia, 44, 129–146 (2001). (2) R. D. Semba, E. J. Nicklett, and L. Ferrucci, Does accumulation of advanced glycation end products contribute to the aging phenotype?, Journals Gerontol. - Ser. A Biol. Sci. Med. Sci., 65 A, 963–975 (2010). (3) R. Nagai, C. M. Hayashi, L. Xia, M. Takeya, and S. Horiuchi, Identification in human atherosclerotic lesions of GA-pyridine, a novel structure derived from glycolaldehyde-modified proteins, J. Biol. Chem., 277, 48905–48912 (2002). (4) D. R. Sell and V. M. Monnir, Structure elucidation of a senescence cross-link from human extracellular matrix. Implication of pentoses in the aging process, J. Biol. Chem., 264, 21597–21602 (1989). (5) K. Tsuji-Naito, H. Saeki, and M. Hamano, Inhibitory effects of Chrysanthemum species extracts on formation of advanced glycation end products, Food Chem., 116, 854–859 (2009). (6) J. W. Baynes and S. R. Thorpe, Glycoxidation and lipoxidation in atherogenesis, Free Radic. Biol. Med., 28, 1708–1716 (2000). (7) S. I. Yamagishi, S. Maeda, T. Matsui, S. Ueda, K. Fukami, and S. Okuda, Role of advanced glycation end products (AGEs) and oxidative stress in vascular complications in diabetes, Biochim. Biophys. Acta - Gen. Subj., 1820, 663–671 (2012). (8) X. Li, SIRT1 and energy metabolism SIRT1 is a Cellular Metabolic Sensor, Acta Biochim. Biophys. Sin., 45, 51–60 (2013). (9) D. Piomelli, A fatty gut feeling, Trends Endocrinol. Metab., 24, 332–341 (2013). (10) S. J. Mitchell, A. Martin-Montalvo, E. M. Mercken, H. H. Palacios, T. M. Ward, G. Abulwerdi, R. K. Minor, G. P. Vlasuk, J. L. Ellis, D. A. Sinclair, J. Dawson, D. B. Allison, Y. Zhang, K. G. Becker, M. Bernier, and R. de Cabo, The SIRT1 activator SRT1720 extends lifespan and improves health of mice fed a standard diet, Cell Rep., 6, 836–843 (2014). (11) Z. Z. Chong, Y. C. Shang, S. Wang, and K. Maiese, SIRT1: New avenues of discovery for disorders of oxidative stress, Expert Opin. Ther. Targets, 16, 167–178 (2012). (12) E. L. Jacobson, P. U. Giacomoni, M. J. Roberts, G. T. Wondrak, and M. K. Jacobson, Optimizing the energy status of skin cells during solar radiation, J. Photochem. Photobiol. B Biol., 63, 141–147 (2001). (13) T. Katayoshi, T. Nakajo, and K. Tsuji-Naito, Restoring NAD+ by NAMPT is essential for the SIRT1/ p53-mediated survival of UVA- and UVB-irradiated epidermal keratinocytes, J. Photochem. Photobiol. B Biol., 221, 112238 (2021). (14) D. G. Dyer, J. A. Dunn, S. R. Thorpe, K. E. Bailie, T. J. Lyons, D. R. McCance, and J. W. Baynes, Accumulation of Maillard reaction products in skin collagen in diabetes and aging, J. Clin. Invest., 91, 2463–2469 (1993). (15) M. D. Farrar, Advanced glycation end products in skin ageing and photoageing: what are the implications for epidermal function? Exp. Dermatol., 25, 947–948 (2016). (16) A. Takahashi, Y. Takabatake, T. Kimura, I. Maejima, T. Namba, T. Yamamoto, J. Matsuda, S. Minami, J. Y. Kaimori, I. Matsui, Matsusaka, F. Niimura, T. Yoshimori, and Y. Isaka, Autophagy inhibits the accumulation of advanced glycation end products by promoting lysosomal biogenesis and function in the kidney proximal tubules, Diabetes, 66, 1359–1372 (2017). (17) S. Radjei, M. Gareil, M. Moreau, E. Leblanc, S. Schnebert, B. Friguet, and C. Nizard, The glyoxalase enzymes are differentially localized in epidermis and regulated during ageing and photoageing, Exp. Dermatol., 25, 492–494 (2016).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)