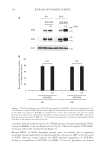
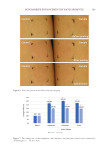
237 J. Cosmet. Sci., 73, 237–244 (July/August 2022) Address all correspondence to Georgios Stamatas at gstamata@its.jnj.com Comparison of Mildness of Six Baby Cleansers Using a Noninvasive Method Combined with Computational Modeling GEORGIOS STAMATAS, HEQUN WANG, ELEA GREUGNY, JALIL BENSACI, ANNA HADER, SIMARNA KAUR AND THIERRY ODDOS Johnson & Johnson Santé Beauté France, Issy-les-Moulineaux, France (G.S., E.G., J.B., T.O.) Johnson & Johnson Consumer Inc., Skillman, New Jersey, USA (H.W., A.H., S.K.) Accepted for publication August 19, 2022. Synopsis The mildness of six baby cleansing products was assessed using a method that combines noninvasive measurement of skin barrier function in adults with predictive computational modeling. This method compares the amount of penetration of a topically applied caffeine solution, used as a marker, on skin sites pretreated with cleansing products. While the clinical part is performed on adults, transferring the caffeine permeation data to an agent-based computational model of the epidermis allows for predictions on infant skin. Thus, cleansing products could be tested and classified based on their predicted effects on infant skin barrier. Overall, this noninvasive method takes less time than other tests, allows for product testing on adults rather than infants, and shows a wide dynamic range to distinguish among skincare products. INTRODUCTION The skin barrier performs essential functions that include retaining water to maintain proper hydration, inhibiting the penetration of microbes and/or allergens, thermoregulation, and gas exchange (1). The presence of a healthy skin barrier is of primary importance during infant skin maturation. Although the infant epidermis possesses a competent skin barrier (2), it differs from that of adults in several ways. Its outermost layer, the stratum corneum, is about 30% thinner compared with that of adults (3). Infant corneocytes and granular cells are smaller and exhibit more rapid turnover than those in adults (3,4). In addition, the water handling properties of the stratum corneum are markedly different between infants and adults (5,6). Infants have significantly higher skin conductance and transepidermal water loss (TEWL), indicating greater hydration yet a less effective skin barrier compared with adults (5,6). These effects were observed across different geographic locations and ethnic groups (5). Water transport resistance profiles in the infant stratum corneum show
238 JOURNAL OF COSMETIC SCIENCE reduced values compared with adults, further indicating a less efficient function of the stratum corneum as a barrier for water permeability in infants (7). Recommendations from pediatricians and dermatologists suggest that appropriately formulated, mild skincare products that do not disrupt the skin barrier should be used on infant skin, especially while infant skin continues to develop (8). While cleansing is important for hygiene, certain surfactants in skin cleansers may be harsh and damage the infant skin barrier, leading to irritation. Thus, it is important to be able to assess the mildness of products aimed for use on infant skin. However, there are concerns with direct clinical testing on infants, especially when this involves certain methods such as exaggerated wash protocols (9,10). A method of evaluating product mildness in adults was developed that involves noninvasively assessing the amount of penetration of a topically applied marker (caffeine) with in vivo confocal Raman microspectroscopy following treatment with a cleanser product (11). The caffeine penetration method has been compared with the more commonly used exaggerated patch and exaggerated arm wash tests for five different formulations in adult volunteers. The ordered rating of the product mildness of the caffeine penetration method agrees with the results of the exaggerated tests, confirming the validity of the method (12). The caffeine permeation data in adults can then be used in a validated, agent- based, computational model of the infant epidermis to predict the penetration of the marker (and therefore the effect of the product) on the infant skin barrier (13). Here, this process is applied for the first time to compare the predicted mildness of six baby cleanser products. METHODS PARTICIPANTS Adult participants included women and men 23–36 years of age with Fitzpatrick skin types II–IV. Exclusion criteria included having a pre-existing dermatologic or chronic medical condition, sunburn, scars, scratches, or active dermal lesion that could interfere with the study or pose a health risk to the participant. In addition, those who were pregnant, planning to become pregnant in the next 30 days, or breastfeeding were excluded, as were those with known allergies or sensitivities to any study ingredient, common topical skincare products, adhesives, or products containing caffeine. STUDY DESIGN This study was conducted at the sponsor’s site in the United States in accordance with the Declaration of Helsinki. The study protocol was reviewed and approved by an institutional review board (study number CS2019-22488), and each participant gave informed consent. The tested products were selected as baby cleansers marketed in the United States and therefore are expected to be generally mild. The ingredients relating to the surfactant system in each product grouped by type of surfactant are given in Supplemental Table S1. All products were tested on 11 volunteers. Three randomized forearm sites of healthy-looking skin were used per volunteer. The tests took place during two visits per volunteer (three products per visit). The two visits took place on nonconsecutive days, and the products were randomized to the tested sites and visits. Participants were asked not to apply any products on either of their forearms the night before or the morning of the study. On the day of the
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)