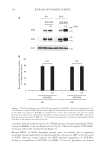
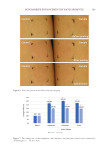
241 NONINVASIVE COMPARISON OF BABY CLEANSER MILDNESS correlation coefficient R2 = 0.98, p 0.01). At first sight, the adult data would be sufficient to order the products according to their mild effect on the skin. However, the relationship is not perfectly linear and there is a certain amount of stochasticity that reflects the stochastic variations in cell proliferation, differentiation, and desquamation introduced in Figure 3. Comparison of the effect of two different cleansers on skin barrier efficacy to external penetration. (A) and (B): Simulation of caffeine penetration following treatment with cleansers A and B, respectively. Caffeine concentration is indicated as gradations of brown color, with darker color corresponding to higher concentration values. (C) and (D): The total caffeine amount penetrating in the stratum corneum (blue) or the whole of the epidermis (orange) for cleansers A and B, respectively. SC, stratum corneum. Figure 4. Predicted penetration of caffeine following treatment with various cleanser products using the infant skin model. Predictions fall into three groups: harsh (product A), moderate (products B, C, D), and mildest (products E, F). Error bars show standard deviation. SC, stratum corneum. *p 0.05 vs. product F.
242 JOURNAL OF COSMETIC SCIENCE the model. Moreover, transferring the experimental data to the infant epidermis model (that recapitulates the stratum corneum and the supra papillary epidermis thickness of infant skin) makes the caffeine penetration results relatable to an infant skin architecture. Therefore, given that the tested products are marketed for infant skin, the model becomes relevant. Protocols on adult skin involving exaggerated repeated arm wash or patch application over 24 hours are often used to assess cleanser irritation potential (9,10). The effect of products on skin barrier function is typically assessed by measuring the rate of TEWL (14). However, TEWL measurements can be variable and can be influenced by the ambient temperature and humidity (14,15). Moreover, TEWL is assessing the inside-out skin barrier function (barrier preventing internal water loss) and not the outside-in function (barrier preventing external penetration). In general, there is a lack of suitable clinical tests that can assess mildness of products for use on infants. To address this need, we introduced a noninvasive method and used it in the present study to compare the mildness potential of six marketed infant cleanser products. This technique noninvasively measures the penetration of a small molecule (caffeine) through the stratum corneum using in vivo confocal Raman microspectroscopy, and has been shown to be consistent with the 24-hour patch test followed by TEWL in adults (12). This method provides caffeine penetration data that can be used as input to a validated, agent-based model of the infant epidermis, resulting in a predicted caffeine penetration profile representative of infant skin. The use of this method in the present study further establishes the usefulness of this method in assessing mildness of cleanser products in adult as well as infant skin. CONCLUSION This validated process enables testing of baby cleansers on adults, while rendering the results relevant to infant skin. The method takes less time compared with other exaggerated tests, is noninvasive, and shows a better dynamic range to distinguish among formulations, particularly mild ones, compared with other commonly used models. ACKNOWLEDGMENTS Writing assistance was provided to the authors by Jennifer L. Giel, PhD, on behalf of Evidence Scientific Solutions, Inc., and was funded by Johnson & Johnson Consumer Inc., Skillman, NJ. This study was funded by Johnson & Johnson Consumer Inc., Skillman, NJ. REFERENCES (1)L . S. Telofski, A. P. Morello, M. C. Mack Correa, and G. N. Stamatas, The infant skin barrier: can we preserve, protect, and enhance the barrier? Dermatol. Res. Pract., 2012, 198789 (2012). (2) J. W. Fluhr, R. Darlenski, A. Taieb, J. P. Hachem, C. Baudouin, P. Msika, C. De Belilovsky, and E. Berardesca, Functional skin adaptation in infancy—almost complete but not fully competent. Exp. Dermatol., 19, 483–492 (2010). (3) G. N. Stamatas, J. Nikolovski, M. A. Luedtke, N. Kollias, and B. C. Wiegand, Infant skin microstructure assessed in vivo differs from adult skin in organization and at the cellular level. Pediatr. Dermatol., 27, 125–131 (2010).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)