243 NONINVASIVE COMPARISON OF BABY CLEANSER MILDNESS (4) J. W. Fluhr, G. Bellemère, C. Ferrari, C. De Belilovsky, G. Boyer, N. Lachmann, C. P. McGuckin, N. Forraz, R. Darlenski, B. Chadoutaud, P. Msika, C. Baudouin, and G. Pellacani, Age-dependent transformation of skin biomechanical properties and micromorphology during infancy and childhood. J. Invest. Dermatol., 139, 464–466 (2019). (5) M. C. Mack, M. R. Chu, N. K. Tierney, E. Ruvolo Jr., G. N. Stamatas, N. Kollias, K. Bhagat, L. Ma, and K. M. Martin, Water-holding and transport properties of skin stratum corneum of infants and toddlers are different from those of adults: studies in three geographical regions and four ethnic groups. Pediatr. Dermatol., 33, 275–282 (2016). (6) J. Nikolovski, G. N. Stamatas, N. Kollias, and B. C. Wiegand, Barrier function and water-holding and transport properties of infant stratum corneum are different from adult and continue to develop through the first year of life. J. Invest. Dermatol., 128, 1728–1736 (2008). (7) M. D. A. van Logtestijn, E. Dominguez-Hüttinger, G. N. Stamatas, and R. J. Tanaka, Resistance to water diffusion in the stratum corneum is depth-dependent. PLoS One, 10, e0117292 (2015). (8) U. Blume-Peytavi, M. J. Cork, J. Faergemann, J. Szczapa, F. Vanaclocha, and C. Gelmetti, Bathing and cleansing in newborns from day 1 to first year of life: recommendations from a European round table meeting. J. Eur. Acad. Dermatol. Venereol., 23, 751–759 (2009). (9) M. W. S. Ong, F. A. Simion, and H. I. Maibach, “Methods for testing irritation potential,” in Textbook of Hand Eczema, A. Alikhan, J. M. Lachapelle, H. Maibach, Eds. (Springer, Berlin, Heidelberg, 2014), pp. 233–246. (10) F. A. Simion, “In vivo models of skin irritation,” in Irritant Dermatitis, A. L. Chew, H. I. Maibach, Eds. (Springer, Berlin, Heidelberg, 2006), pp. 489–499. (11) G. N. Stamatas and E. Boireau-Adamezyk, Development of a non-invasive optical method for assessment of skin barrier to external penetration. Presented at the Biomedical Optics and 3-D Imaging Meeting Congress, 28 April–2 May 2012, Miami, FL, USA (2012). (12) G. N. Stamatas, E. Boireau-Adamezyk, and T. Oddos, In vivo assessment of cleanser mildness by confocal Raman microspectroscopy. Presented at the International Investigative Dermatology Annual Meeting, 16–19 May 2018, Orlando, FL, USA (2018). (13) G. N. Stamatas, J. Bensaci, E. Greugny, S. Kaur, H. Wang, M. V. Dizon, M. J. Cork, A. J. Friedman, and T. Oddos, A predictive self-organizing multicellular computational model of infant skin permeability to topically applied substances. J. Invest. Dermatol., 141, 2049–2055.e1 (2021). (14) H. Alexander, S. Brown, S. Danby, and C. Flohr, Research techniques made simple: transepidermal water loss measurement as a research tool. J. Invest. Dermatol., 138, 2295–2300.e1 (2018). (15) K. Grice, H. Sattar, and H. Baker, The effect of ambient humidity on transepidermal water loss. J. Invest. Dermatol., 58, 343–346 (1972).
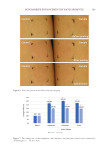
244 JOURNAL OF COSMETIC SCIENCE SUPPLEMENTAL DATA Supplemental Figure S1. Caffeine penetration data from the simulation on the infant epidermis model correlate with data from the clinical experiment on adults (R2 = 0.98, p 0.01). Supplemental Table S1 List of ingredients comprising the surfactant systems of the different baby cleanser products tested. Type of surfactant Surfactant Product Amphoteric (zwitterionic character) Cocamidopropyl betaine (CAPB) C, D, E, F Sodium cocoamphoacetate C Disodium cocoamphodiacetate C Non-ionic PEG-80 sorbitan laurate D, F Glyceryl oleate D PPG-2 hydroxyethyl cocamide F Coco glucoside C, D, E Decyl glucoside D, F Caprylyl/capryl glucoside B Cationic Quaternium-80 C Polyquaternium-10 C Anionic Sodium lauroyl methyl isethionate B Sodium methyl cocoyl taurate F PEG-150 pentaerythrityl tetrastearate F Sodium lauroyl oat amino acids B Sodium lauroyl glutamate E Sodium laurate E Sodium methyl lauroyl taurate E Sodium methyltaurate E Potassium olivate A Potassium cocoate A PEG, polyethylene glycol.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)