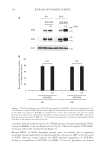
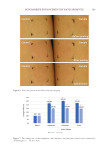
211 Skin Barrier Enhancement by Saccharomyces in 10 minutes. In addition, the result of colorimetric measurements showed that sample removed redness by 7.26% (p 0.05) in 10 minutes. So, the yeast essence balance lotion containing SRFF showed an immediate repairing capacity after sun exposure. According to the 28 days of clinical treatment, on the skin where yeast essence balance lotion was applied, on average, the whiteness of skin increased by 20.13% (p 0.05) and the skin gloss increased by 17.39% (p 0.05). Because of the promotion effect of SRFF on TJ expression, the yeast essence balance lotion containing SRFF showed long-term protection and repair after sun exposure. Overall, our results demonstrate that SRFF can increase skin barrier function by increasing the expression of TJs, such as claudin-1, claudin-4, and occludin. Our previous paper discussed the antiaging effect of SRFF in cosmetic products (1). This paper further explains its effect on skin barrier function, as well as its application in yeast essence balance lotion to reduce photoaging and its synergy with sunscreen products. CONCLUSION SRFF is a TJ expression promotion and sun-protection synergy active ingredient. Using the same sunscreen, the skin treatment side with the yeast essence balance lotion containing SRFF showed brightness, whiteness, and gloss increase, so the lotion has a significant synergistic anti-photoaging effect. Overall, our results demonstrated that SRFF can increase skin barrier function against sunlight by increasing the expression of TJs. The induction of TJs by SRFF also benefits skin health in the aspects of antiaging in the short and long terms. Clinical testing proves the antiaging effect of the yeast essence balance lotion with SRFF. REFERENCES (1) F. Yang, Z. Zhou, M. Guo, and Z. Z. Zhou, The study of skin hydration, anti-wrinkles function improvement of anti-aging cream with alpha-ketoglutarate, J. Cosmet. Dermatol., 21(4), 1736–1743 (2022). (2) M. Svoboda, Z. Bílková, and T. Muthný, Could tight junctions regulate the barrier function of the aged skin? J. Dermatol. Sci., 81, 147–152 (2016). (3) E. Kohl, J. Steinbauer, M. Landthaler, and R. M. Szeimies, Skin ageing, J. Eur. Acad. Dermatol. Venereol., 25, 873–884 (2011). (4) H. Shah and S. R. Mahajan, Photoaging: new insights into its stimulators, complications, biochemical changes, and therapeutic interventions, Biomed. Aging Pathol., 3, 161–169 (2013). (5) K. Bäsler, S. Bergmann, M. Heisig, A. Naegel, M. Zorn-Kruppa, and J. M. Brandner. The role of tight junctions in skin barrier function and dermal absorption, J. Control. Release, 242, 105–118 (2016). (6) G. Krause, L. Winkler, S. L. Mueller, R. F. Haseloff, J. Piontek, and I. E. Blasig, Structure and function of claudins, Biochim. Biophys. Acta, 1778(3), 631–645 (2008). (7) K. Turksen and T. C. Troy, Barriers built on claudins, J. Cell Sci., 117, 2435–2447 (2004). (8) M. Furuse, T. Hirase, M. Itoh, A. Nagafuchi, S. Yonemura, S. Tsukita, and S. Tsukita, Occludin: a novel integral membrane protein localizing at tight junctions, J. Cell Biol., 123(6 Pt 2), 1777–1788 (1993). (9) J. Ikenouchi, M. Furuse, K. Furuse, H. Sasaki, S. Tsukita, and S. Tsukita, Tricellulin constitutes a novel barrier at tricellular contacts of epithelial cells, J. Cell Biol., 171(6), 939–945 (2005). (10) K. Ebnet, A. Suzuki, S. Ohno, and D. Vestweber, Junctional adhesion molecules (JAMs): more molecules with dual functions? J. Cell Sci., 117, 19–29 (2004).
212 JOURNAL OF COSMETIC SCIENCE (11)C . F. Hung, C. L. Fang, S. A. Al-Suwayeh, S-Y. Yang, and J-Y Fang, Evaluation of drug and sunscreen permeation via skin irradiated with UVA and UVB: comparisons of normal skin and chronologically aged skin, J. Dermatol. Sci., 68(3), 135–148 (2012). (12) T. Yuki, A. Hachiya, A. Kusaka, P. Sriwiriyanont, M. O. Visscher, K. Morita, M. Muto, Y. Miyachi, Y. Sugiyama, and S. Inoue, Characterization of tight junctions and their disruption by UVB in human epidermis and cultured keratinocytes, J. Invest. Dermatol., 131(3), 744–752 (2011). (13) T. Yamamoto, M. Kurasawa, T. Hattori, T. Maeda, H. Nakano, and H. Sasaki, Relationship between expression of tight junction-related molecules and perturbed epidermal barrier function in UVB- irradiated hairless mice, Arch. Dermatol. Res., 300(2), 61–68 (2008). (14) K. Biniek, K. Levi, and R. H. Dauskardt, Solar UV radiation reduces the barrier function of human skin, Proc. Natl. Acad. Sci. U. S. A., 109, 17111–17116 (2012). (15) S. Tsukita and M. Furuse, Claudin-based barrier in simple and stratified cellular sheets, Curr. Opin. Cell Biol., 14(5), 531–536 (2002). (16) M. Furuse, M. Hata, K. Furuse, Y, Yoshida, A. Haratake, Y. Sugitani, T. Noda, A. Kubo, and S. Tsukita, Claudin-based tight junctions are crucial for the mammalian epidermal barrier: a lesson from claudin-1- deficient mice, J. Cell Biol., 156(6), 1099–1111 (2002). (17) J. A. Tunggal, I. Helfrich, A. Schmitz, H. Schwarz, D. Günzel, M. Fromm, R. Kenler, T. Krieg, and C. M. Niessen, E-cadherin is essential for in vivo epidermal barrier function by regulating tight junctions, EMBO J., 24(6), 1146–1156 (2005). (18) J. C. Jones and R. D. Goldman, Intermediate filaments and the initiation of desmosome assembly, J. Cell. Biol., 101(2), 506–517 (1985). (19) N. Kirschner, R. Rosenthal, M. Furuse, I. Moll, M. Fromm, and J. M. Brandner, Contribution of tight junction proteins to ion, macromolecule, and water barrier in keratinocytes. J. Invest. Dermatol., 133(5), 1161–1169 (2013). (20)C . V. Itallie, C. Rahner, and J. M. Anderson, Regulated expression of claudin-4 decreases paracellular conductance through a selective decrease in sodium permeability, J. Clin. Invest., 107(10), 1319–1327 (2001). (21) N. Sonoda, M. Furuse, H. Sasaki, S. Yonemura, J. Katahira, Y. Horiguchi, and S. Tsukita, Clostridium perfringens enterotoxin fragment removes specific claudins from tight junction strands: evidence for direct involvement of claudins in tight junction barrier, J. Cell Biol., 147(1), 195–204 (1999). (22)C . Stremnitzer, K. Manzano-Szalai, A. Willensdorfer, P. Starkl, M. Pieper, P. König, M. Mildner, E. Tschachler, U. Reichart, and E. Jensen-Jarolim, Papain degrades tight junction proteins of human keratinocytes in vitro and sensitizes C57BL/6 mice via the skin independent of its enzymatic activity or TLR4 activation, J. Invest. Dermatol., 135(7), 1790–1800 (2015). (23) T. Yuki, H. Yoshida, Y. Akazawa, A. Komiya, Y. Sugiyama, and S. Inoue, Activation of TLR2 enhances tight junction barrier in epidermal keratinocytes, J. Immunol., 187(6), 3230–3237 (2011). (24) M. Malminen, V. Koivukangas, J. Peltonen, S-L. Karvonen, A. Oikarinen, and S. Peltonen, Immunohistological distribution of the tight junction components ZO-1 and occludin in regenerating human epidermis, Br. J. Dermatol., 149(2), 255–260 (2003). (25) E. J. O’Keefe, R. A. Briggaman, and B. Herman, Calcium-induced assembly of adherens junctions in keratinocytes, J. Cell Biol., 105(2), 807–817 (1987).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)