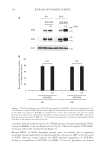
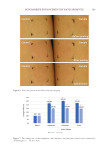
190 J. Cosmet. Sci., 73, 190–200 (July/August 2022) Address all correspondence to Kentaro Tsuji-Naito, knaito@dhc.co.jp Sirtuin-1 Prevents Advanced Glycation End Products From Accumulating in Epithelial Keratinocytes TAKESHI KATAYOSHI, NATSUKO KITAJIMA, TAKAHISA NAKAJO AND KENTARO TSUJI-NAITO DHC Corporation, Division 2, Mihama-ku, Chiba, Japan (T.K., N.K., T.N., K.T.-N.) Accepted for publication July 18, 2022. Synopsis Accumulation of advanced glycation end products (AGEs) in skin occurs because of photo-aging rather than chronological aging. However, it is unclear how ultraviolet (UV) radiation affects AGE deposition in the short term. In this study, keratinocytes were exposed to AGE-bovine serum albumin (BSA) as exogenous AGEs and evaluated the intracellular AGE-BSA accumulation. The intracellular AGE-BSA levels in UVA/B- exposed cells were significantly higher than those in unexposed cells, whereas the residual AGE-BSA levels in the medium were unchanged. The autophagy inhibitors increased the subcellular AGE-BSA levels. As reported previously, the intracellular nicotinamide adenine dinucleotide (NAD+) levels dropped after UVA/B irradiation. Because the NAD+-dependent enzyme sirtuin-1 (SIRT1) positively regulates autophagy, UVA/B- induced NAD+ depletion may suppress the SIRT1-mediated degradation of exogenous AGEs, leading to AGE-BSA accumulation. One SIRT1 inhibitor, EX527, clearly increased the subcellular AGE-BSA levels, whereas SIRT1 activators and an NAD+-boosting agent, nicotinamide mononucleotide, decreased them. These results support the idea that SIRT1 plays a key role in preventing exogenous AGE accumulation. Furthermore, UVA/B irradiation or EX527 significantly influenced the mRNA expression of glyoxalase 1 and glyoxalase 2, which metabolize reactive AGE precursors. Our study showed that SIRT1 is a potential target for anti-aging approaches against AGE deposition in photo-aged skin. INTRODUCTION Advanced glycation end products (AGEs) is a collective term for products that are nonenzymatically formed through Maillard reactions, wherein the carbonyl groups of reducing sugars react with proteins and lipids (1). AGEs accumulate in various parts of the body through aging, which causes cells and organs to decrease in function (2). Diverse AGEs with specific characteristics such as fluorescence, brownish color, and crosslinking have been identified (3–5). Several studies have indicated that accumulation of AGEs exacerbates pathological conditions of degenerative diseases (6), including diabetes, atherosclerosis, chronic renal failure, and Alzheimer’s, and it causes vascular complication in diabetes (7).
191 Accumulation of Advanced Glycation End Products Sirtuin-1 (SIRT1) serves as an intracellular sensor for nicotinamide adenine dinucleotide (NAD+) concentration and regulates energy metabolism through its NAD+-dependent deacetylase activity (8). In recent years, impairment in SIRT1 activity has been implicated in the pathogenesis of aging-associated diseases (9). Enhanced expression and activity of SIRT1 has been reported to extend the life span of experimental animals and cells via its anti-aging effects (10,11) therefore, focus has been laid on activating SIRT1 during aging. Ultraviolet (UV) irradiation has been shown to decrease intracellular NAD+ levels in the skin (12,13). This suggests that impairment of SIRT1 activity more profoundly proceeds in photo-exposed areas than in photo-protected ones. Several reports have implied that photo-aging accelerates AGE accumulation in the skin (14,15). However, the causal relationship between such accumulation and SIRT1 is unknown. Herein, we show that AGE accumulation induced by UV exposure is caused partly by reduced activity of SIRT1. MATERIALS AND METHODS MATERIALS We purchased an anti-AGE antibody (KH001) from the Medical Chemistry Pharmaceutical Company (Hokkaido, Japan). An anti-beta actin antibody (ab8226), EX527, and bafilomycin A1 were obtained from Abcam (Cambridge, MA, USA). Chloroquine was purchased from Tokyo Chemical Industry (Tokyo, Japan). An anti-bovine serum albumin (BSA) antibody (66201-1-lg) was obtained from Proteintech (Rosemont, IL, USA). SRT1720 was purchased from MedChemExpress (Monmouth Junction, NJ, USA). Nicotinamide mononucleotide (NMN) was purchased from Oriental Yeast (Tokyo, Japan). Resveratrol was purchased from Sigma-Aldrich (St. Louis, MO, USA). We purchased a horseradish peroxidase (HRP)- conjugated anti-mouse antibody from GE Healthcare (Chicago, IL, USA). PREPARATION OF AGE-BSA AGE-BSA was prepared as previously described in the literature (16). Briefly, BSA was incubated with 0.5 M d-glucose in 0.4 M PBS at 37°C for 8 weeks. Free glucose was removed using Amicon centrifugal filters with a 50 kDa molecular weight cutoff device (Merck Millipore, Darmstadt, Germany). The concentration of AGE-BSA was calculated using the Pierce BCA protein assay kit (Thermo Fisher Scientific). CELL CULTURE Normal human epidermal keratinocytes (NHEKs newborn/male, Thermo Fisher Scientific, Waltham, MA, USA) were cultured in an EpiLife® medium containing 60 µM calcium supplemented with human keratinocyte growth supplement (both from Thermo Fisher Scientific) at 37°C under a humidified atmosphere with 5% CO 2 . The medium was changed every other day until the culture attained approximately 50% confluence, after which it was changed every day. The cells were passaged when they attained 80–90% confluence. NHEKs were not used beyond passage four.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)