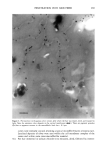
336 JOURNAL OF COSMETIC SCIENCE oxidized sulfinic acid, -SO-OH (1073 cm ']) or sulfonic acid, SO2 -OH (1041 cm']). In contrast, the more moist ambient prevalent at 60-65% RH (Fig. 2b), leads to the domination of what is tentatively proposed to be sulfinic acid. Another possible assignment is the monoxide. More importantly, in photochemical oxidation of hair, unlike chemical approaches to the same, sulfonic acid is not the end product. There also appears to be a dormant period during photolysis under more humid conditions before photoproduct formation begins (sulfinic acid production, Fig. 2b). 0.5 0.4- 122g cm '• [ Piedmont 10-14% RH 1167 '• 1111 cm • • ,•,,•.•,•, A • /// 100 2o0 300 4OO 5OO Duration of Irr.diatlon (h) Piedmont 80.-66% RH 0,5 -e- 1229 cm 't II /I-'- •o7• •m -- 0.2 o. 1 .. .• , 0 0 -• 0 100 2OO 30O 400 Duration of Irradiation (h) (a) (b) Fig. 2 Kinetics of product formation during photolysis of Piedmont hair under low (a) and high (b) ambient humidities In summary, it is suggested that photochemical oxidation of the disulfide moiety in hair proceeds initially through formation of thiosulfonate. Under dry conditions of irradiation, this is the dominant product. Under more humid conditions, this lower oxide of sulfur is further oxidized to sulfinic acid. This contrasts with chemical oxidation of hair wherein cysteic acid is the dominant product though lower sulfur oxides could also be present. References 1. J. Nachtigal and C. Robbins, Text. Res. ,L., 40, 454 (1970) 2. C.J. Dixon and D. W. Grant, Photochemistry andPhotobiology, 18, 387 (1973) 3. U. Schumacher-Hamedat, J. F6hles and H. Zahn, Proc. 7 th Int. Wool Text. Res. Cvnf, Tokyo, Japan, IV, 120 (1985) 4. C.M. Pande, d. Soc. Cosmet. Chern., 45, 257 (1994) 5. V. Signori and D. M. Lewis, int. d. Cosmet. Sci., 19, 1 (1997) 6. N. Kharasch and C. Y. Meyers, Eds., The Chemistry of Organic Sulfur Compounds, Vol. 2, Chap. 15. Pergamon Press (1966)
2001 ANNUAL SCIENTIFIC SEMINAR 337 CURDLAN: STYLE MEMORIZING POLYMER FOR HAIR FIXATIVE Katsunori Yoshida, Ph.D., Daigo Mizumoto, M.S., Tomoyuki Kawasoe, M.S., Yoshiharu Tsuji, M.S., Masaaki Uemura, M.S. and Toshio Yanaki, Ph.D. Shiseido Research Center, 2-2-1 Hayabuchi, Tuzuki, Yokohama, 224-8558, Japan Introduction A variety of synthetic and natural polymers have been developed as a hair fixative to meet various consumers' needs. One of the most popular materials is vinyl polymer, particularly, the copolymer of vinylpyrrolidone. Despite its superior ability to fixate hairstyle, the application feeling of the polymer is uncomfortably hard and tough. To improve the application feeling, some of natural polymers and their derivatives are proposed. In general, however, the ability of natural polymers to keep hairstyle nnder high humidity is unsatisfactory. Thus, a hair fixative polymer that gives soft and light application feeling but provides high humidity resistance is desired. In this •vork, we propose the use of curdian, a natural occurring polysaccharide,• as a novel hair fixative polymer that bears both soft application feeling and high humidity resistance. Experimental [- cu:oH ca.oa • o o purchased from Takeda Chemical Industry (Tokyo, ' , , [ Japan), and used as received. The chemical structure of k ' oH , o• 3,, curdian is shown in Figure 1. Figure 1. Chemical structure of curdian. Rheological measurements. Polymer films were prepared by drying their solutions at 25øC in clean bench for overnight. The curdlan film was prepared as the same manner' as described above except by drying its gel that was prepared by heating curdian sol at 80 øC for 30 min. The theological properties of films were measured by a dynamic viscoelastic an•alyzer RSA II (Rheometric Scientific F.E., Tokyo). Scanning electron microscopy (SElVl). Hair was dipped in polymer solution, and the excess amount of solution was combed out. The hair was dried at 80 øC for ,30 min, and the polymer film on hair surface was studied by SEM (S-4200, Hitachi, Tokyo). Humidity resistance ability (R %).2 Polymer solutions (0.5%, 0.4 g) were applied to hair tresses (160 mm long, 1 g). The hair tresses were curled and then dried at 80 øC for 30 min. The curled hair was hung in incubator (40 øC, 75% RH). The length of curled hair was measured in time course. The humidity resistance ability(R%) was evaluated by folloxving equation: R%= Lø 'Lt xlO0 (1) L o - L•
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)