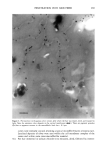
344 JOURNAL OF COSMETIC SCIENCE LIPID NANOTUBES AS SKIN PENETRATION MODULATORS Vitthal Kulkarni, Ph.D., Joretta Wong, Duncan Aust, Ph.D., James Wilmott and James Hayward, Ph.D. The Collaborative Group, Ltd., 3 Technology Drive, East Setauket, NY 11733 vitt haL kulkarni @ c ollabo. com INTRODUCTION Human skin is constantly exposed to the harsh environment including bright sun light, atmospheric pollutants and extreme wet or dry conditions that cause premature skin aging and may lead to derreal conditions. Topical supplement of skin care actives is necessary to maintain a healthy skin. To get the actives safely into skin there needs to be a delivery system that can carry the actives efficiently and be non toxic to skin. Advantages of a suitable "delivery system" include the possibility of reduced toxicity, controlled release, and deep penetration of an active into skin (Kulkami et al., 2001 a, 2001b). In cosmetic formulations it is equally important to retard or prohibit the penetration of actives such as sunscreens. Sphingolipids are an important class of lipids that have unusual thermotropic properties than the normal glycerol based phospholipids and play very important role in structure and function of cell membranes. Recent investigation has revealed that certain sphingolipids including cerebrosides and ceramides form tubular microstructures upon hydration (Kulkami et al. 1999). However, the tubular microstructures have found limited application in the delivery systems (Zarif et al. 2000a 2000b). An appropriate combination of phospholipids, sphingolipids and certain other lipids form tubular microstructures that have shown promising applications in the topical delivery systems as penetration enhancers. Our recent studies indicate that depending on the mode of application of nanotubes, they can promote or retard skin penetration significantly. Here we report our studies on lipid nanotubes and their applications in cosmetic delivery systems as controllers of skin penetration. MATERIALS AND METHODS Lipid nanotubes were formed with proprietary high-pressure high-shear technique. Retinol palmitate was incorporated into the tubes as an active ingredient. A nano-dispersion of retinol palmitate was also prepared with established procedure. A panel of five volunteers was chosen to test the products "Nanotubes retinol palmitate" and "nano-dispersion retinol palmitate" both at the same concentration (0.5%). Known amount of test product was applied to the forearm of volunteers at four different places. At desired time points after application, the applied surface area was rinsed with ethanol to recover un- penetrated active. Retinol palmitate from skin washings was quantitatively analyzed via HPLC. In a separate study we applied a solution of nanotubes for 30 min (at 37øC) to a model skin epidermis equivalent to human skin, (Epi 606 MatTek, Inc.) followed by incubation with a nano-dispersion of oil which also contained a fluorescent dye octadecyl filuorescene (ODF) for 2 hrs at 37øC. The model skin was then examined in a confocal microscope to determine the depth of penetration of the marker dye ODF. RESULTS AND DISCUSSION * '• b•r = 1 un• , , 0 • 4 6 8 "Hn'•, he Figure 1: A: Transmission electron micrograph of lipid nanotubes. B: Retinol palmitate penetrated deep into skin. Curve I represents nanotube formulation and curve 2 represents the nano-dispersion
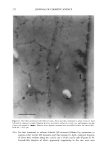
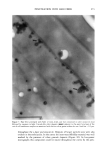
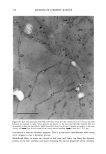
2001 ANNUAL SCIENTIFIC SEMINAR 345 Figure 1A shows the transmission electron micrograph of typical nanotubes which are several microns long and diameter ranges from 30-60 nm. The figure lB shows the amount of retinol palmitate penetrated in the skin over time. Nearly 70% of retinol from nanotubes penetrated into skin while only 48% of retinol palmitate penetrated into skin from a nano-dispersion suggesting that nanotubular structures enhanced the penetration. This experiment suggests that when an active is incorporated into a nanotubular microstructures it may enhance the penetration. Our data suggests that mechanism of penetration of nanotubes is different than that of oil-in-water dispersion. A possible reason for enhanced penetration by nanotubes is that the lipid composition of nanotubes is compatible with skin lipid composition and also the diameter of nanotubes is in the same range of skin pore size. It is also reported that the lipid tubular microstructures are more stable in harsh environment like surface of the skin than the liposomes or oil-in- water dispersions. In a separate study we applied blank nanotubes to a model skin epidermis for 30min then the skin was rinsed off with a buffer solution and applied a solution of nano-dispersion containing ODF in oil phase. The confocal microscopy revealed that application of nanotubes almost entirely blocked the penetration of ODF (see Fig. 2) suggesting that nanotubes probably formed a mesh like coating on the skin which did not let the oil-in water dispersion to penetrate in the epidermis. Conclusions Figure 2: Orthogonal view of reconstructed fluorescent images from optical slices of model epidermis (Epi 606 from MatTek, Inc.) acquired using a confocal microscope. Left: Blank model epidermis shows auto fluorescence arising due to the lipids present in the skin. Middle: 10% EFA-nano-dispersion for 2hr (37øC) the intense fluorescence indicates that ODF penetrated to 10-12•tm deep in the skin. Right: Treated the skin with blank nanotubes for 30 min (37øC) then incubated with 10%-EFA-nano-dispersion for 2hrs (37øC). The fluorescence is at the same level as that of the background indicating that ODF did not penetrate in this case suggesting that the nanotubes blocked the penetration of EFA-nano-dispersion. Retinol palmitate from a nanotubular formulation showed 20% more penetration in skin than from a nano-dispersion formulation over 6hrs of time period In-vitro studies showed that the marker dye ODF incorporated in a nano-dispersion did not penetrate in the epidermis that was treated with nanotubes Our studies suggest that depending on the method of application, the nanotubular microstructures may enhance or retard the rate penetration of an active in skin. References Kulkarni, V. S., Aust, D., Wilmott, J., and Hayward, J. A. (2001a) "Liposomes In Cosmetics: An Overview Of Production, Characterization, And Applications" The Manufacture and Chemist• of Cosmetics, Allured Publ. Corp., Illinois, --in press Kulkarni, V. S., Aust, D., Wilmott, J., and Hayward, J. A. (200lb) "Cosmetic Delivery Systems: An Overview" The Manufacture and Chemistry of Cosmetics, Allured Publ. Corp., Illinois, --in press Kulkarni, V.S.J.M Boggs, and R.E. Brown (1999) "Modulation of nanotube formation by structural modification of sphingolipids" Biophys. J. 77, 319-330 Zarif, L., and Mannino, R. J. (2000a) "Cochleates. Lipid-based vehicles for gene delivery-concept, achievements and future development" Adv. Exp. Med. Biol. 465:83-93 Zarif, L., Graybill, J. R., Periin, D., Najvar, L., Bocanegra, R., and Mannino, R. J. (2000b) "Antifungal activity of amphotericin B cochleates against Candida albicans infection in a mouse model" Antimicrob. Agents Cheroother. 44(6): 1463-1469
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)