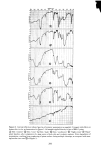
344 JOURNAL OF THE SOCIETY OF. COSMETIC CHEMISTS 0 I 2 3 4 5 6 7 HOURS Figure 4. Two different runs at 0.5 % sodium lauryl sulfate into the concentrated surfactant solution above the membrane (osmotic effect). This flow can be calculated to be something on the order of 1 to 2 mg/hr/cm 2, and is a negli- gible factor for the time and concentrations described below. PERMEATION OF SURFACTANTS Seemingly contradictory opinions have appeared in the literature concerning the permeability of mammalian stratum corneum to surfactants and ionic species in general. The viewpoints of Blank (7,8), that both cationic and anionic surfactants penetrate human epidermis with great difficulty or not at all, and of Scala (9), that permeability of human skin continually increases with exposure to these surfactant types, were reconciled by Scheuplein (5) who showed the great effect of concentration. The data for sodium lauryl sulfate given below confirm his interpretation.
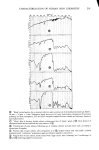
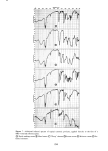
PERMEATION OF KERATINOUS SUBSTRATES 345 As a preliminary it may be noted that considerable barrier strength does exist in the stratum corneum against nonionic and cationic surfactants. Thus the cationic, myristyl- dimethylbenzyl ammonium chloride (Barquat MS-100) applied at the level of 10% to neonatal rat stratum corneum requires about six days for penetration. Figure 5 shows the permeability curve, which resembles the schematic of Figure 2. Using eq 1 above, one can calculate from the slope an apparent permeability, P, of 2.2 x 10 -6 cm/sec, and from the lag time of six days D = 0.8 x 10 -• cmg/sec. The permeability value is un- reasonably high since it is even greater than that for water (see above). If one now uses these values of P and D in eq 3 to calculate the partition coefficient K, a value of 1380 is obtained. For the surfactant concentration of about 0.1 g/cm a this implies a concentration of 138 g/cm a of Barquat MS-100 in the swollen skin, a physical impossi- bility. It is plausible that sometime between three and six days the membrane has been structurally altered to permit such a high permeation. Since this change may well be non-uniform throughout the membrane, there is little point in the calculation of P or D in such a case. The same phenomenon was found for the nonionic Tergitol 15-S-9 at m- z o 80 70 60- 40- 30- 20- 10- ß 6 8 DAYS 0 2 4 10 12 Figure 5. Permeability of Barquat MS- 100 at 10% concentration 14
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)