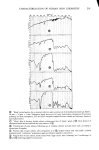
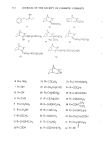
ISOCAMPHANE DERIVATIVES Nummer Chemische Bezeichnung Literatur 49 50 51 52 53 54 55 56 3,3-Dimethyl-bicyclo [2.2.1 ] hept- 2-yl-methylbromid lIsocamphenilanylbromidl 5-13,3-Dime thyl -bicyc lo [2.2. ! ]- hept-2-yliden}-2-methyl-pent-2-enol liso-Santaloll 1-13,3-Dimethyl-bicyclo[2.2.1 ]- hep t-2-yl}-4 -methyl-pent -2- enon 5-[3-Methyliden-2-methyl-bicyclo- [2.2.1]hept-12-yl}-2-methyl-but-2-enol l[3-Santaloll 5-13,3-Dimethyl-bicyclo[2.2.1 ]- hept-2-yl}-2-methyl-pent-2-enol {6 -Santalol} 1 -{3,3-Dimethyl-bicyclo[2.2.1 ] hept- 2-yl}-4-methyl-pentanon 3-{2'-Furyl}-l-{3,3-dimethyl-bicyclo- [2.2.1 ]hept-2-yl }-prop-2-enon 3-Cyclohexyl- 1 -{3,3-dimethyl-bicyclo- [2.2.1 ]hept-2-yll-propanon 21 23 24 21 1 2 3 4 •H2OH • 5 10 1• /••COCH• 15 /••c H(OH)C Hi 16 14 •CH2
314 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS 21 22 •CH2C H2C HC(CH3)2 ••11 HCHC H2 OH C HC H2C HC(C H3)C H2 OH 50 O••COCH3 24 H3C CCH2 ••CHC &5 •H2C H2C HC(C H 3) C H2 OH 52 H2CHC(CH3)2 !• R= NH 2 7 R=OH • R=CN 9 R=CHO •1 R=CH2NH2 12 R=CH20H q? R=COCH] q9 R=CH(OH)CH] 20 R=COOH 23 R= COC2H 5 25 R =CH2CH2CHO 26 R= CH2N(CH]) 2 27 R= C(CH3)20H 28 R=CH(OH)C2H 5 29 R=COOCH] ]0 R=OCOCH] 31 R=CH2NO 2 ]2 R =COCHCHCH] 33 R=CHCHCOCH3 :]4 R=COC]H7 35 R=CHCHCOOH 36 R =CON(CH3)2 /CH:] 38 R=C(OH)Nc2H 5 ]9 R =CH(OH)C]H7 4O R=CH2CH2CO0 H 41 R= CH2N•(CH3) 2 0 42 R- Br
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)