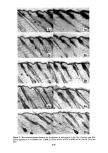
340 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS EXPERIMENTAL Stratum corneum of neonatal rats was used as a model for human skin. Live young rats one or two days old were obtained from Marland Breeding Farms, West Milford, New Jersey. The animals were sacrificed by being placed in an atmosphere of CO2 for several hours after death the whole skin was removed by a surgical scalpel. The skin was placed in a desiccator jar and exposed to ammonia vapor for 1 to 3 hr. Following this, the skins were put in water and the epidermal layer was gently separated from the dermis. The epidermis so obtained was floated on the surface of a pan of water. After an hour the membrane was removed by bringing up a metal screen under it. The membrane was placed top down on a wet paper towel and the screen removed. At this point the Malpighian layer could be gently scraped off, leaving the desired stratum cor- neum on the towel. The paper and stratum corneum were placed again in water until separation occurred. The stratum corneum layer was recovered by a small Teflon screen and air-dried. Isolation of the stratum corneum follows a method outlined for us by E. J. Singer and E. Bolsits of Lever Brothers Co., Edgewater, New Jersey. A typical piece of stratum comeurn was about 25/•m thick and 5 x 6 cm in area. It weighed about 20 rag, corresponding to a density of 0.7. The permeability cell used was modeled after a description by Loveday (12), and is shown schematically in Figure 1. The temperature of the experiments was that of the laboratory, 23øC + iøC. The surfactants used in this study were: Tergitol 15-S-9 (Union Carbide Corp.), the 9 mol ethoxylate of a secondary C H to C •a alcohol. PERMEABILITY CELL AFTER LOVEDAY (1961) SOLUTION- SKI N --STIRRER BAR Figure 1. Schematic drawing of permeability cell
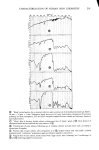
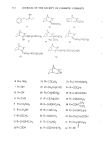
PERMEATION OF KERATINOUS SUBSTRATES 341 Barquat MS-100 (Lonza, Inc.), myristyldimethylbenzyl ammonium chloride. Sodium lauryl sulfate (SLS) was obtained as a pure white crystalline powder from BDH Chemicals. Analyses of the first two materials were made spectrophotometrically, by absorption at 337 nm for Tergitol and at 262 nm for Barquat. In these cases, the small amount of protein leached from the stratum comeurn does not interfere with absorption peaks of the surfactants. Radiotagged material was purchased from Amersham-Searle (Des Plaines, IL) in the form of small individual ampoules. Each ampoule contained 2.47 mg of SLS with an activity of 100 microcuries. The tag was present as the S-35 isotope and is thus in the anionic part of the surfactant. Solutions of desired concentration were made up of the nonradioactive powder and one ampoule was added with stirring. For permeability ex- periments a small sample (0.1 g) of the water in the lower part of the cell was removed by pipette and put in a 1-oz counting vial filled with the scintillant liquid, Instagel. Ra- dioactivity was determined by scintillation counting in a Packard 3255 Counter. For sorption experiments skin samples of about 2 mg each were placed in 20 ml of tagged solution for various set times, removed and rinsed twice for a few seconds with distilled water to remove entrained solution. The skin was then dissolved in UNISOL and a scintillant cocktail added. Radioactivity was again determined in the Packard Counter. Polymer JR is a quaternary nitrogen-containing cellulose ether (13). The JR-400 grade was used for the experiments reported here. Its approximate molecular weight is 400,000. NOTE ON CALCULATIONS For •biological membranes such as skin, whose thickness is difficult to measure, it is convenient to work with the apparent permeability defined by (14) Q/A = P Ac t, [1] where Q is the amount of solute which penetrates through area A in time t P is the ap- parent permeability constant and Ac is the difference in concentration between the two sides of the membrane. For this work Ac is taken to be simply the initial concentration of the upper solution, since the concentration in the stirred water is always close to zero. An idealized Q vs. time curve is shown in Figure 2. There is an initial slow growth of Q leading to a straight line portion, the extrapolation of which back to the time axis gives the "lag" time, To. From this time a diffusion constant, D, can be calculated by (14) D = h2/6To [2] where h is the thickness of the swollen membrane. The plot illustrates how both P and D can be derived from permeability data. For purposes of calculation h has been assumed here to be 50/•, which is about twice the dry thickness of the stratum cor- neum. (It should be appreciated that individual membranes vary in thickness and that the swollen thickness changes slowly with time.) In the ideal case, the apparent permeability is related to the diffusion constant, D, by the equation:
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)