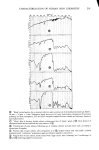
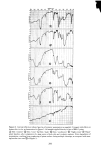
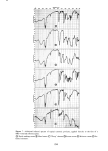
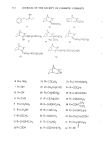
SORPTION OF KERATINOUS SUBSTRATES 337 increases in the presence of added salt but decreases in the presence of added nonionic surfactant. Lauryl ether sulfates are sorbed to a lesser extent than SLS and their uptake decreases with ethylene oxide content. By comparison of sorption data obtained by radiometric and gravimetric techniques, it has been demonstrated that a simple weighing technique can be employed for measur- ing the uptake of surfactants and simple salts, in view of their relatively high sorption values. REFERENCES (5) (6) (7) (8) (9) (10) (11) (12) (13) (14) 15) 16) (17) (18) (19) (1) G. V. Scott, C. R. Robbins and J. D. Barnhurst, Sorption of quaternary ammonium surfactants by hair, J. Soc. Cosmet. Chem., 20, 135 (1969). (2) P. Finkelstein and K. Laden, The mechanism of conditioning of hair with alkyl quaternary ammonium compounds,Appl. PolymerSymp., No. 18, 673 (1971). (3) M. F. Nelson, Jr. and D. Stewart Jr., The adsorption of N-acyl sarcosines on various protein materials, J. Soc. Cosmet. Chem., 7,122 (1956). -• (4) S. P. Harrold and B. A. Pethica, Thermodynamics of the adsorption of small molecules by proteins, Trans. Faraday Soc., 54, 1876 (1958). I. H. Blank and E. Gould, Penetration of anionic surfactants into skin, J. Invest. Dermatol., 33,327 (1959). H. E. Garrett, Detergents--what they are and how they work, Trans. St. John Hosp. Dermat. Soc., 51, i66 (i965). J. A. Faucher and E. D. Goddard, Sorption of a cationic polymer by stratum corneum, J. Soc. Cosmet. Chem., 27,543 (1976). E.J. Singer, P. C. Wegmann, M.D. Lehman, M. S. Christenson and L.J. Vinson, Barrier development, ultrastructure, and sulfhydryl content of the fetal epidermis, J. Soc. Cosmet. Chem., 22, 119 (1971). P. B. Sram and H. J. White,Jr., The application of radiochemical techniques to the study of the interac- tion of hair fibers with aqueous solutions, Text. Res. J, 24, 785 (1954). J. A. Medley and M. W. Andrews, The effect of surface barrier on uptake rates of dye into wool fibers, Textile Res. J, 29,398 (1959). J. A. Medley and M. W. Andrews, The kinetics of wool dyeing, Text Res. J., 30, 855 (1960). N.H. Leon, Structural aspects ofkeratin fibers, J. Soc. Cosmet. Chem., 23,427 (1972). W. C. Preston, Some correlating principles of detergent action, J. Phys. Chem., 52, 84 (1948). M. Abu-Hamdiyyah and K. J. Mysels, The dialysis of sodium dodecyl sulfate, J. Phys. Chem., 71,418 (1967). J. Griffith, "The Uptake of Sodium Dodecyl Sulfate by Wool," Proc. 3rd Internat. Congr. Surface Activ., Cologne, 1960, Vol. 4, pp 28-36. J. C. Chen, The Interactions of an Anionic Surfactant with Keratin Fibers, Ph.D. Thesis, Princeton University, Princeton, New Jersey, 1971. I. J. Lin, "Hydrophobic Properties of Long-Chain Ionic Surfactants," in Colloid and Interface Science, Vol II, M. Kerker, Ed., Academic Press Inc., New York, New York, 1976, pp 431-443. M. J. Schick and D. J. Manning, Micelie formation in mixtures of nonionic and anionic detergents, J. Am. Oil Chem. Soc., 43, 133 (1966). A. W. Finkstein, "Eye Irritation Studies on Some Common Shampoo Surfactants," paper presented at the Annual Scientific Meeting of the Society of Cosmetic Chemists, New York, New York, December 1976. (20) J. Clifford and B. Sheard, Nuclear magnetic resonance investigation of the state of water in human hair, Biopolymers, 4, 1057 (1966). (21) R. L. Anderson, J. M. Cassidy, J. R. Hansen and W. Yellin, Hydration of stratum corneum, Biopolymers, 12, 2789 (1973). (22) C. A. Nelson, The binding of detergents to proteins, J. Biol. Chem., 246, 3895 (1971).
J. Soc. Cosmet. Chem., 29, 339-352 (May 1978) Interaction of keratinous substrates with sodium lauryl sulfate.. II. permeation through stratum corneum J. A. FAUCHER and E. D. GODDARD Union Carbide Corporation, Tarrytown, NY 1059I. Received September 9, 1977. Synopsis Neonatal rat STRATUM CORNEUM was used as a model membrane to investigate PERMEATION through mammalian skin. Passage of materials through these membranes was determined by use of radio- tagged compounds and by spectrophotometric analysis. The anionic surfactant SODIUM LAURYL SULFATE penetrates the stratum corneum even at low concentrations. The diffusion constant for this process is about 10 -•ø cm"/sec, compared to 10 -6 cm"/sec for free diffusion in water. This SURFACTANT is bound to the skin in large amounts, up to 50% by weight at high concentrations. Pretreatment of the membrane by a cationic cellulose polymer (which is itself strongly sorbed) greatly reduced the amount of surfactant which passed through the membrane. INTRODUCTION Considerable study has been made in the past of the effect of surfactants on the permeability of mammalian skin. For example, Bettley and Donoghue (1) showed that soap not only penetrates the skin barrier, but also makes the barrier more permeable to water and other solutes. A number of studies thereafter emphasized the increased per- meation of water or salts (2-6). Relatively little, however, has appeared on the permea- tion of surfactants themselves through skin. Perhaps the first quantitative study was by Blank and Gould in 1959 (7) showing that anionic surfactants in low concentration penetrated human epidermis with difficulty. Blank's subsequent work on a cationic surfactant indicated virtually no penetration at all (8). With somewhat greater con- centrations, Scala et al. (9) and Howes (10) demonstrated more permeation by various anionic surfactants. All of these previous studies were quite limited in terms of the range of concentrations studied and the time scale investigated yet both factors have been specifically recognized to be of great importance in the permeation process (5). The authors' interest in the permeation of surfactants through skin arose in con- nection with a clinical study of antiirritation effects of a cationic polymer (11). A detailed study of both sorption and permeation of the anionic surfactant sodium lauryl sulfate has been made with the aim of clarifying the mechanism of action of these ob- served protective effects. 339
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)