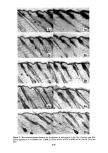
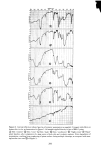
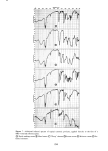
Figure 6. Internal reflection infrared spectra of cosmetic preparations as supplied. Compare with effects on human skin, in situ, as characterized in Figure 5. All samples applied directly to face of KRS-5 prism. (•) Skin "cleanser" (• Skin "toner" ¸ Skin "mask" (• Skin "conditioner" (•)"Night cream" (•)"Blank" prism baseline, after completion of a large series of both skin and cosmetic analyses. Note degradation of transmission, resulting from roughening of prism surface during multiple cleanings, as compared with start- ing baseline (see trace (• of Figure 5). 293
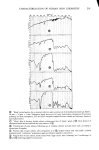
294 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS depth" of the electromagnetic sensing waves, is a consequence of use of the cosmetics. The cosmetics applied assisted in the removal of rough, loosely attached stratum cor- neum fragments and also improved the refractive index matching across the prism/skin interface by leaving the residual components of the formulations. It is significant to note, here, as will be confirmed in later figures, that no apparent change in the skin "moisture" level i• demonstrated after this cosmetic treatment. The second lowest trace of Figure 5 characterizes once again the forearm skin of our volunteer, but now after further treatments with a "skin mask" cosmetic preparation and a skin "condi- tioner" (according to the suppliers' instructions for the intended use of these products). This infrared spectrum reveals, with the exception of some residual hydrocarbon and fatty-ester components imbibed into the epidermal layers, the return of the skin quality to, essentially, its pretreated state. This suggests that the polyol-containing"mask" and "conditioner" formulations did little besides remove excess hydrocarbon and (seem- ingly weakened) epidermal layers affected by the "cleanser" and "toner" formulations. Finally, the lowest infrared spectrum included in Figure 5 characterizes the forearm skin of our subject as treated with a "night cream" after its "cleansing" and "condition- ing" according to the cosmetic supplier's instructions. As the spectra collected in Figure 6 show, "moisturizing" cream has the highest fatty-ester content of ahy of the cosmetics used in this series. It is, in fact, these fatty esters which are taken up into the skin to soften, lubricate, or plasticize it. The only indication of a truly increased "mois- ture" content (that is, additional water retention) of the skin is the slight skewing of the N-H absorption band in the lowest trace of Figure 5. This band, normally symmetrical about 3300 cm -•, is shifted towards the 3400-cm -• absorption maximum which characterizes the O-H resonance of retained hydroxyl or water groups. The continued observation of intense absorption bands in the Amide I and Amide II region of the spectrum illustrates the "night cream" to have been taken up into the epidermal layers rather than to be lying in a thin film above them. In the latter case, even a film thickness of a few microns would significantly damp the analytical sensitivity to proteinaceous layers beneath them. Some judgment of the actual influence of a number of cosmetic products on human skin in situ, examined without distress to the volunteer subject and in a rapid, noninva- sive manner, is thus easily accomplished by the internal reflection, infrared spectro- scopic technique. The second important feature of the method, beyond its ability to evaluate skin before and after its modification, is the method's suitability (without modification) for the direct analysis of the cosmetic preparations themselves. The tech- nique does not require their dilution, extraction, or complex processing in any manner beyond that experienced in their normal application to the skin. Figure 6 collects internal reflection, infrared spectra of neat cosmetic preparations (as supplied and ap- plied directly to human skin with the outcome characterized in Figure 5). All of the samples were applied directly to the face of the same KRS-5 prism used in recording the skin spectra. The top trace of Figure 6 characterizes the skin "cleanser" formula- tion. Scanning from left to right across this trace, it is seen that the major infrared bands are for hydrocarbon components at 2950 and 2850 cm-• fatty esters, at 1740 cm-• confirming absorptions for hydrocarbons at 1470 and 1380 cm-• and confirming absorptions for the ester components at 1280, 1180 and 1130 cm -•. A final confirming absorption band for the long chain, aliphatic hydrocarbons present in this formulation appears at 740 cm -•. On the basis of numerous similar analyses, primarily of pure known compounds and their mixtures, the top spectrum in Figure 6 would be compati-
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)