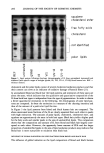
194 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS 0.03 f Na/TEA 0.025 0.02- pte o.o15 , O.Ol 0.005 o o lO 20 30 40 50 60 Surfactant Concentration, mra Figure 10. Binding of surfactants to HSC, one-minute treatment at 37C. to the stratum corneum, then the obvious question is: Why does the binding increase above CMC in some cases (see Figures 2-7)? In this regard, a brief discussion on the structure of stratum corneum itself will be helpful. According to the currently accepted model, stratum corneum has a biphasic structure, with protein-rich corneocytes em- bedded in a matrix of lipids. This is analogous to a brick-and-mortar structure, with the corneocytes and the barrier lipids representing the bricks and the mortar, respectively (32). The lipids consist of both fluid and rigid lipids. The corneocyte envelope also consists of membrane proteins and lipids together with some covalently attached surface lipids, which makes them compatible with the surrounding matrix lipids (30). Thus, for the surfactant molecules to interact with the deeper protein-rich regions in a normal corneum, they must diffuse through the lipid regions. It is believed that the network structure of the stratum corneum and the conformational features of the membrane proteins limit the accessibility of the surfactant to all the potential binding sites on the keratinous proteins. This may involve a thermodynamic component arising from the cohesive forces holding the network together, including those from the conformational features of the proteins. In addition, a kinetic component related to the slow diffusion of surfactant to the binding sites, especially at the corneocyte-lipid matrix interface, also may be affecting the binding behavior. Micelies may contribute to surfactant binding in several ways that include: i) solubilization of fluid lipids leading to exposure of binding regions that were otherwise unexposed, ii) abstraction of calcium or other multivalent ions to reduce corneocyte adhesions, iii) introducing osmotic stresses on the membrane leading to its breakdown, and iv) contributing to favorable free-energy changes that accompany the cooperative binding process leading to protein unfolding. The latter is equivalent to direct binding of surfactant aggregates, such as the conventional micelies or premicellar clusters (33), to proteins. Processes i and ii enhance the accessibility of the proteins in the lower regions of the stratum corneum and are essentially kinetic in
BINDING OF SURFACTANTS 195 character. Processes iii and iv are intrinsic effects of micelies to the binding equilibrium of insoluble proteins and are, thus, thermodynamic in nature. These contributions are discussed below. It has been suggested that surfactants above their CMC cause some delipidization of the stratum corneum (5,34), even though no consensus exists on this topic (35-37). An alternative view is that surfactants can remove selectively certain components such as the ceramides and in the process cause changes in the lipid composition (38). It is possible that the reasons for the discrepancies existing in the literature are essentially due to the differences in the protocols used in various studies, especially the surfactant concentra- tion and time and frequency of exposure. Our unpublished results of an ongoing TEM study of porcine stratum corneum treated with SDS for one hour, similar to the con- ditions of the binding experiments, indicate that SDS at levels above the CMC cause significant delipidization (39). Similarly, our ultrastructural studies of human skin subjected to an in vitro arm-wash protocol reported elsewhere showed that soaps caused significant lipid removal and protein damage (40). Such removal of lipids by surfactant micelies may play an important role in enhancing the diffusion of surfactant monomers and micelles to the corneocyte-matrix interface. Therefore, continued binding above the surfactant CMC is not unreasonable, even when only monomers are involved in the binding process. Note that the results of surfactant binding to delipidized corneum discussed earlier (see Figure 4) may appear to be contradictory to the proposed hypoth- esis of micelies enhancing the monomer binding by enhancing the surfactant diffusion by removing free lipids. The apparent contradiction can, however, be reconciled by noting that the delipidization study discussed earlier was done using solvents and that subsequently the skin was dried before contacting with the surfactant solution. During drying the covalently linked lipids would have formed a strong hydrophobic network that would be much more impenetrable to the surfactants than the normal corneum containing both the free lipids and the covalently bonded lipids. Yet another mechanism by which anionic micelies can influence the binding process may be by sequestering metallic ions such as calcium. Even though the role of ions such as calcium in stratum corneum cohesion is not well understood, it is known that addition of EDTA, a well-known metal ion chelator, enhances the surfactant-induced release of corneocytes (41-42). If the underlying reason for the effect of EDTA is metal ion complexation, one can expect micelies also to play a similar role and reduce the cohesion between corneocytes, which in turn can enhance the penetration of surfactants to surface regions of corneocytes. Once the surfactant molecules obtain access to protein-rich corneocyte regions, the extent of binding and especially cooperative binding in the postmicellar region is primarily determined by the inherent tendency of the surfactant monomer to bind to the proteins in the corneocyte membrane. The cooperative binding in these systems may well induce the changes in the conformation of the keratinous proteins present in the corneocyte membrane, which in turn may lead to damage to the corneum. This may require some threshold level of binding to overcome the cohesive forces arising from the conformation of the protein as well as the lipids holding the membrane together. This is similar to Breuer's concept of the "elastic energy" holding the keratinous network together (26). Surfactant binding obviously induces repulsive electrostatic forces that at some critical binding should overcome "cohesive forces" and lead to an unfolding of the protein. Thus, while all micelies, including nonionic surfactant micelies, can cause some
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)