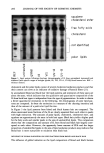
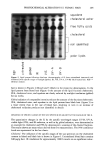
196 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS removal of fluid lipids from the matrix lipid, significant damage to the protein itself will still depend upon the ability of the surfactant monomer to bind to the corneocyte membrane proteins. This is consistent with the reported observations that nonionic surfactants such as Triton X-100, in combination with some mechanical agitation, can release corneocytesrfrom the lipid matrix without damaging the membrane envelope (43). In contrast to this, treatments with harsh surfactants such as SDS or soaps result in damage to lipids and corneocyte proteins (38,39). In addition to the above mechanisms, micelles may be contributing directly to the unfolding process by creating osmotic stresses on the corneocyte envelope. It is well known that corneocytes swell when exposed to water and aqueous surfactant solutions (44). In general, the extent of corneum swelling by anionic surfactants increases with their harshness toward skin (44). It has also been reported that the extent of swelling as a function of SDS concentration attains a plateau value near the CMC of SDS (44), suggesting that monomers that bind to the corneum, including the accessible sites on the membrane, may be enhancing corneocyte swelling. This is consistent with the present observation that initial surfactant binding to the corneum exhibits a near plateau or a slowly rising region around the surfactant CMC. Interestingly, it has also been reported (44) that the surfactant-induced swelling process is reversible up to about 35 mM (1%) SDS, but is not reversible above about 70 mM (2%), indicating irreversible damage at such levels. The sharp rise in the SDS binding beyond the CMC also occurs around 60 mM, suggesting that this rise is responsible for the irreversible damage to the corneum. The origin of the sharp increase in SDS binding occurring around 60 mM may involve contributions from miceliar exclusion and the associated excluded volume effects. Be- yond the CMC, the corneocyte envelope may act as a semipermeable membrane that does not allow the micelles to penetrate into the cells. This is similar to Middleton's (45,46) concept that the corneocyte membrane acts as a semipermeable envelope holding the water-absorbing NMFs (natural moisturizing factors) within the cell and allowing water to move across the membrane. Exclusion of micelles will indeed result in an osmotic effect due to an increase in the concentration of micelles, leading to mechanical stresses on the membrane. In normal systems, one would have expected deswelling of the corneum because of osmotic stresses. Interestingly, the reported results do not indicate any deswelling above the surfactant CMC (44). This may be because the water respon- sible for the swelling is associated with the hydration of the surfactant-bound proteins and the NMFs, and the removal of the bound water may require a critical pressure (energy) that is greater than that which can be achieved by micellar exclusion. However, another pathway is available to release the osmotic stress. This may involve unfolding of the membrane proteins that are already weakened by surfactant binding, leading to a physical breakdown of the membrane structure. The unfolded proteins, in turn, expose more sites for surfactant binding. This phenomenon can also be viewed in the following manner: When surfactant binds to water-swollen proteins in a cooperative fashion, micelles are essentially transferred from solution to the "solid" protein phase. This process leads to a decrease in the chemical potential associated with the micelles. If this chemical potential difference, coupled with the free energy of cooperative surfactant- protein binding, is greater than the molecular interactions holding the protein network intact, the protein should unfold to accommodate bound micelles. Thus an increase in
BINDING OF SURFACTANTS 197 binding occurs above the CMC. Obviously the situation is complex, and a quantitative model is required to sort out the details. All of the four factors discussed earlier are in line with the results obtained for the kinetics of SDS binding to human and guinea pig stratum corneum. In both these cases, the binding was found to increase with increase in time up to about six hours. Super- imposed over this increase, there exists a sharply rising region at SDS levels well above the CMC. As mentioned earlier, the relatively mild SLI, on the other hand, does not exhibit such a marked increase in binding above its CMC. The relative magnitudes of different miceliar contributions to enhanced surfactant bind- ing above the CMC are not clear at present. Binding experiments with already-dispersed corneocytes as well as those with barriers that are damaged by techniques such as thermal pretreatment or sonication may provide better insight into the relative contributions. EFFECT OF pH ON SLI BINDING As mentioned earlier, SLI binding was found to exhibit a minimum around pH 7-9, with an increase in both the acidic and alkaline regions. This behavior can be explained in terms of the dependence of properties of the stratum corneum proteins and lipids on pH. An underlying assumption in the following analysis is that "true equilibrium" is difficult to attain in a complex system such as stratum corneum and that the reported binding at different pH values may be at different stages of equilibrium depending upon such factors as thermodynamic binding potential, accessibility to different binding sites, etc. An increase in pH can be expected to decrease the binding of the anionic surfactant to keratin. This is because keratin has isoelectric points at about pH 5 (47). Above this pH, the net negative charge on the protein can be expected to increase with increase in pH. This, in turn, will lower the thermodynamic driving force for surfactant binding to the protein. The experimentally observed decrease in binding with increase in pH up to about a pH of 9.0 is consistent with this expectation. However, the binding appears to increase above pH 9. This is possibly due to the effect of pH on matrix lipids as well as the membrane lipids. In general, the aqueous solubility of the fatty acids, which are a significant component of the bilayer lipids in the stratum corneum (5), can be expected to increase with increase in pH, especially at pH values above 8 or 9. Even though the ionization of the carboxylic acid group can occur at pH values above 5, formation of an acid-soap complex limits the solubility of the soap below pH 9. Above this pH, a marked increase in solubility with pH can be expected. Thus, the delipidization process itself may be more favorable at pH values above 9. Furthermore, the negative charge on the protein structure itself can be expected to increase with increase in pH, and this may reduce the conformational stability of the folded protein. Thus the overall accessibility and vulnerability of the structure to surfactants can be expected to increase eventually with increase in pH. This cutoff seems to be around pH 9 and becomes pronounced at pH values 10. The effect of pH on the mildness of formulated skin cleansing compositions is complex and still a matter of debate (see reference 48 for a review of the subject). For formulations containing potentially ionizable components, Murahata and Aronson recently showed that increase in pH can have a pronounced effect on mildness (48). The above discussion
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)