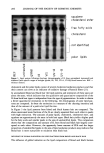
292 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS REFERENCE (1) G. E. Pierard and C. Pierard-Franchimont, Dihydroxyacetone test as a substitute for the dansyl chloride test, Dermato/ogy, 185, 133-137 (1993). Characterization and chemistry of enhanced-efficacy (activated) aluminum-zirconium-glycine (AZG) antiperspirant actives' A new approach ALLAN H. ROSENBERG, Summit Research Labs, Huguenot, NY 12746, INTRODUCTION Enhanced-efficacy (activated) antiperspirant actives were first introduced into the mar- ketplace in the early 1980s in the form of activated aluminum chlorohydrate powder (ACH), which was primarily used in aerosol products. The key factors for manufacturing these materials were the ability to characterize the aluminum polymer distributions present in the actives by chromatographic and metal analysis techniques and the ability to achieve an aluminum polymer distribution that gives enhanced efficacy compared to the standard (normal) ACH powders (1-3). Figure 1 shows aluminum polymer distributions for standard and activated ACH actives obtained from a Sephadex G-25 (S) column. Since larger polymers elute faster than smaller polymers, the molecular weight of the polymers decreases as elution time (i.e., Kd values) increases. Notice that activated ACH contains a large amount of the alu- minum polymer designated as A1 Kdo.4, while standard ACH contains little AI Kdo. 4. As seen from many clinical efficacy studies, the enrichment in A1 Kdo. 4 is the primary reason for the superior efficacy of activated ACH compound to standard ACH. •. ss.•m NO• AC44 Z ]f• : • ß -, ACTIVAToeOAC41 Figure 1. Aluminum polymer distributions for normal (standard) and activated ACH actives.
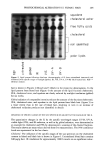
PREPRINTS OF THE 1996 ANNUAL SCIENTIFIC MEETING 293 In the mid 1980s enhanced efficacy (activated) aluminum-zirconium-glycine actives were introduced. Activated AZG powders were also enriched in AI Kdo. 4 (4-6). Thus it was assumed that activated AZG containing large amounts of AI Kdo. 4 should outperform standard (normal) AZG, which contains little AI Kdo. 4. Surprisingly, many so-called activated AZG salts were no better than standard AZG salts and in fact could show inferior performance compared to standard AZG actives. This anomoly becomes clear when one considers the effect of zirconium chemistry on efficacy, which until now has not been addressed. CHARACTERIZATION OF ZIRCONIUM POLYMER DISTRIBUTIONS Using Sephacryl columns in conjunction with ICP techniques to measure Zr, Rosenberg et al. (7) were able to determine Zr polymer distributions present in AZG actives. Thus, changes in Zr polymer distribution as a function of synthesis and processing could be determined and then correlated to clinical efficacy results. A striking example of how Zr polymer distribution affects clinical efficacy is seen in Figure 2, which shows Zr polymer distributions for a so-called activated tetra AZG powder containing large amounts of AI Kdo.4, and for a standard tetra AZG powder containing small amounts of AI Kdo. 4. Clinical results comparing these materials showed that the standard AZG active out- performed the so-called activated AZG (statistically significant result). This result is surprising only if one solely considers AI polymer distributions. However, the larger molecular weight Zr polymer distribution of the so-called activated AZG compared to the standard AZG negatively impacted the efficacy of the activated AZG even though it contained large amounts of the AI Kdo. 4 polymer. Other correlations of Zr polymer distributions versus clinical efficacy confirm that Zr polymer distributions are more important than AI Kdo. 4 enrichment, with lower molecular weight Zr polymer distri- butions being more desirable with respect to enhanced efficacy (7). FACTORS AFFECTING ZR POLYMER DISTRIBUTIONS Since Zr polymer distribution strongly impacts efficacy, it is important to understand how synthesis and processing affect the resultant Zr polymer distribution of the finished AZG product. Some important factors to consider are the nature of the Zr starting materials: heat, addition of glycine, and contact time of the AI and Zr components before spray drying. This approach to manufacturing enhanced efficacy AZG actives is • m NOFU4,q,L A7.G • m m m m m A•TIYAT•D•T O m • O a i/ -..% EL•ION TIME Figure 2. Zirconium polymer distributions for a normal (standard) and a so-called activated AZG.
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)