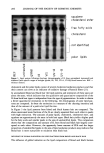
198 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS and the results in Figure 8 indicate that an alkaline pH can also increase the suscepti- bility of the stratum corneum to increased binding of anionic surfactants, which can in turn lead to protein unfolding. This effect is thus likely to also contribute to the harshness of alkaline compositions to the skin. SUMMARY AND CONCLUSIONS 1. TEA-oleate binds to stratum corneum in higher amounts than all other surfactants at low surfactant concentrations. This is consistent with the expected higher surface activity of longer-chain soaps compared to lower-chain-length soaps and surfactants. At concentrations above about 20 mM (for example, at a concentration of 40 mM), the surfactant binding to HSC is about the same for oleate, laurate, and SDS. Importantly, the binding of SLI is significantly lower than that of the other three surfactants. The extent of binding correlates with the irritation potential of these surfactants measured by the zein dissolution test as well as with the well-known clinical irritation of cleansing bars based on these surfactants. 2. Binding of SDS, TEA/Na oleate, and TEA/Na laurate to HSC increases above the surfactant CMC. Similarly, the binding of SDS to guinea pig stratum corneum shows a sharp increase at concentrations well above CMC. The reasons for the increase in surfactant binding above their CMC are not clear at present. The role of micelles in removing lipids and increasing the accessibility of membrane proteins to surfactant monomers may be implicated in the enhanced surfactant binding at concentrations above the CMC. 3. Increasing the temperature from room temperature to 37øC results in an increase in binding of SDS to HSC, with no measurable effect on SLI. 4. The SLI exhibits a pH-dependent binding behavior to HSC, with a minimum around the pH 7-9 region. REFERENCES (1) C. Prottey, Factors which determine the skin irritation potential of soaps and detergents, J. Soc. Cosmet. Chem., 26, 29-46 (1975). (2) G. Imokawa, K. Sumura, and M. Katsumi, Study on skin roughness caused by surfactants: II. Correlation between protein denaturation and skin roughness, J. Am. Oil. Chem. Soc., 52, 484-489 (1975). (3) G. Imokawa and T. Takeuchi, Surfactants and skin-roughness, Cosmet. Toiletr., 91, 32-46 (1976). (4) L. D. Rhein and F. A. Simion, "Surfactant Interactions with Skin," in Interfacial Phenomena in Biological Systems, Surfactant Science Series, Vol. 39, Max Bender, Ed. (Marcel Dekker, New York, 1991), pp. 33-49. (5) W. Matthies, "Dermatological Observations (Humans)," in Anionic Surfactants, Their Biochemistry, Toxicology and Dermatology, 2nd ed., Surfactant Science Series, Vol. 43, C. Gloxhuber and K. Kun- stler, Eds. (Marcel Dekker, New York, 1992), pp. 291-329. (6) J. Steinhardt and J. A. Reynolds, Multiple Equilibria in Proteins (Academic Press, New York, 1969), pp. 234-301. (7) C. Tanford, The Hydrophobic Effect.' Formation of Micelles and Biological Membranes, 2nd ed. (Wiley- Interscience, New York, 1980), pp. 146-164. (8) M. J. Schwuger and F. G. Bartnik, "Interaction of Anionic Surfactants with Proteins, Enzymes and Membranes," in Anionic Surfactants, Surfactant Science Series, Vol. 10, C. Gloxhuber, Ed. (Marcel Dekker, New York, 1980), pp. 1-49.
BINDING OF SURFACTANTS 199 (25) (26) (27) (9) M. N. Jones, Surfactant interactions with biomembranes and proteins, Chem. Soc. Rev., 21, 127-136 (1992). (10) I. D. Robb, "Polymer-Surfactant Interactions," in AnionicSurfactants, Physical Chemistry of Surfactant Action, Surfactant Science Series, Vol. 10, Lucassen-Reyenders, Ed. (Marcel Dekker, New York, 1981), pp. 109-142. (11) K. P. Ananthapadmanabhan, "Protein-Surfactant Interactions," in Interactions of Surfactants with ?oly- mers and Proteins, E. D. Goddard and K. P. Ananthapadmanabhan, Eds. (CRC Press, Boca Raton, Florida, 1993), pp. 319-366. (12) E. Gotte, Skin compatibility of tensides measured by their capacity for dissolving zein, in Proc. 4th Int. Cong. Surface Active Subs., Brussels, 3, 83-90 (1964). (13) F. Swanson, Clinical evaluation of a new neutral detergent bar, J. Am. Med. Assoc., 162, 459-461 (1956). (14) D. D. Strube, S. W. Koontz, R. I. Murahata, and R. F. Theiler, The flex wash test: A test method for evaluating the mildness of personal washing products,J. Soc. Cosmet. Chem., 40, 297-306 (1989). (15) O. Laurie and J. Oakes, Protein-surfactant interactions: Spin label study of interaction of BSA with SDS, J. Chem. Soc. Faraday Trans. I, 72, 1324-1328 (1976). (16) J. Oakes, Protein-surfactant interactions: NMR and binding studies of interactions between BSA and SDS, J. Chem. Soc. Faraday Trans. I, 70, 2200-2209 (1974). (17) M. N. Jones, Biochem. J., 151, 109-114 (1975). (18) K. P.M. Ananthapadmanabhan, M. Aronson, N.J. Turro, and X. Lei, A spectroscopic study of interaction of SDS with bovine serum albumin, Langmuir, 11, 2525-2533 (1995). (19) K. Kerr, Surfactant-protein interactions, Unpublished results, Unilever Research, Port Sunlight, 1990. (20) J. C. Griffith and A. E. Alexander, Equilibrium adsorption isotherms for wool detergent systems, J. Coll. Interface Sci., 25, 311-316 (1967) see alsoJ. ColloidlnterfaceSci. 25, 317-321 (1967). (21) S. P. Harrold and B. A. Pethica, Thermodynamics of the adsorption of small molecules by proteins, Trans. Faraday Soc., 54, 1876-1884 (1958). (22) I. H. Blank and E. Gould, Penetration of anionic surfactants into skin, J. Invest. Dermatol., 33, 327-336 (1959). (23) J. A. Faucher and E. D. Goddard, Interaction of keratinous substrates with sodium lauryl sulfate: Sorption, J. Soc. Cosmet. Chem., 29, 323-338, (1978). (24) R. D. Kulkarni and E. D. Goddard, "Destruction of the Electrophysiological Potential of Excised Frog Skin by Surfactants," in Advances in Bioelectrochemistry.' Ions, Surfaces, Membranes, Adv. Chem. Series, no. 188, M. Blank, Ed. (ACS, Washington, D.C., 1976)pp. 445-459. G. Imokawa and T. Takeuchi, Surfactants and skin-roughness, Cosmet. Toiletr., 91, 32-46 (1976) G. Imokawa, Comparative study on the mechanism of irritation by sulfate and phosphate type anionic surfactants,"J. Soc. Cosmet. Chem., 31, 45456 (1980). M. M. Breuer, The interaction between surfactants and keratinous tissues, J. Soc. Cosmet. Chem., 30, 41-64 (1979). J. G. Dominguez, J. L. Parra, R. M. Infante, F. Pelejero, F. Balaguer, and T. A. Sastre, New approach to the theory of adsorption and permeability of surfactants on keratinic proteins: Specific behavior of certain hydrophobic chains, J. Soc. Cosmet. Chem., 28, 165-182 (1977). (28) A. Conrads and H. Zahn, A study of the interaction of sodium dodecyl sulfate with proteins of human heel stratum corneum, Int. J. Cosmet. Sci., 9, 29-46 (1976). (29) P. D. Wertz, D. C. Swartzendruber, D. J. Kitko, C. Kathi, M. D. Madison, and T. Downing, The role of the corneocyte lipid envelope in cohesion of the stratum corneum, J. Invest. Dermatol., 89, 169-172 (1987). (30) D.C. Swartzendruber, P. W. Wertz, M.D. Madison, and D. T. Downing, Evidence that the corneocyte has a chemically bound lipid envelope, J. Invest. Dermatol., 88, 709-713 (1987). (31) R. I. Murahata, R. Toton-Quinn, and M. Finkey, Effect of pH on the production of irritation in a chamber irritation test, J. Am. Acad. Dermatol., 18, 62456 (1988). (32) P.M. Elias, Epidermal lipids, barrier function and desquamation, J. Invest. Dermatol., 80, 44s-49s (1983). (33) R. L. Hill and L. D. Rhein, Characterization of the sub-micellar species in mixed surfactant solutions. I: Mixtures of sodium dodecyl sulfate and an alkyl polyethoxy sulfate, J. Dispersion Sci. Tech., 9(3), 269-308 (1988).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)