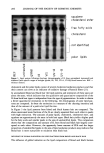
REDUCTION OF HUMAN HAIR 223 0 • -5 Half-life= 123 s Z -6 -- -7 I I I I 0 200 400 600 800 1000 Time (seconds) Figure 5. Amino acid analysis: comparison of the reduction of whole-fiber disulfide bonds in single-head virgin hair by 1 M ammonium thioglycolate, pH 9.4, with 1 M cysteamine, pH 7.6, 23øC, using the mean reaction rate constant for each condition and first half-life. [•, 1 M ATG, pH 9.4, slope = - 5.65 X 10 -3 s-• /•, 1 M cysteamine, pH 7.6, slope = -1.79 X 10 -3 s -• from the SFTK measurements. Both studies indicate that reduction of disulfide bonds by ATG is faster than by cysteamine. The rate of reduction for both treatments was faster from the SFTK measurements, as the hair fibers were reduced under 1.5% strain. The rate constant from SFTK measurements was faster, as only the removal of stress- supporting disulfide bonds is monitored in this measurement. The correlation of results indicates that stress-relaxation data represent not only the removal of stress-supporting disulfide bonds but correlate with disulfide reduction in general. It is possible that the difference in the rates of reaction for ATG and cysteamine may result from the degree of fiber swelling caused by differences in pH of the solutions. The influence of swelling on the condition of the hair has been studied by several authors (2,31,33-38). Hair swelling occurs diametrically and is accompanied by lateral con- traction. The rate and extent of hair swelling is dependent on pH (increasing sharply above pH 9) (5,37) and concentration of the reducing agent (37). However, there is no difference in swelling of hair in the pH range of 8-10 in the absence of reducing agents. Furthermore, Wortman and Souten (39) have shown that length setting does not occur therefore, changes in length from swelling are unlikely. This result indicates that the measured decreases in force can be attributed solely to decreased fiber strength. Thus, it seems likely that rather than "leading to" faster reduction, greater swelling in re- ducing solutions "results from" faster reduction, at least in the pH range of 8-10. Swelling affects both the condition of the hair and the diffusion rate of the reducing agent into the hair. An increase in the degree of swelling causes an increase in the rate of diffusion of the reducing agent and a decrease in the processing time (i.e., rate of reaction). Overall, the percentage of disulfide bonds cleaved is controlled by the pH of the solution and the pK of the reducing agent (34). Therefore, at pH 8.0, cysteamine
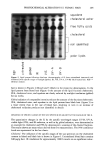
224 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS Z -7 Half-life = 394 s Half-life = 210 s Half-life = 123 s Half-life = 93 s I I I 0 200 400 600 800 1000 Time (seconds) Figure 6. Correlation of the results of amino acid analysis and SFTK measurements: comparison of the reduction of stress-supporting and whole-fiber disulfide bonds in single-head virgin hair by 1 M ammonium thioglycolate, pH 9.4, with 1 M cysteamine, pH 7.6, 23øC, using the mean reaction rate constant for each condition as obtained from SFTK experiments and amino acid analysis. O, SFTK data for 1 M ATG, pH 9.4, slope = -7.44 X 10 -3 s- • I, SFTK data for 1 M cysteamine, pH 7.6, slope = - 3.29 X 10 -3 s-• &, amino acid analysis data for 1 M ATG, pH 9.4, slope = -5.65 X 10 -3 s-• •', amino acid analysis data for 1 M cysteamine, pH 7.6, slope = - 1.79 X 10 -3 s-• reduces hair faster than ATG (see Tables I and II) because the concentration of active species, RS-, is much higher due to the lower pK a of cysteamine. In addition to pH and swelling factors, cysteamine may reduce the disulfide bonds at a slower rate than ATG for other reasons. First, a five-membered ring may form in solution via hydrogen bonding (Figure 7a). Formation of a five-membered ring is possible, as it has been shown (40) that cysteamine exists in the gauche conformation 66% of the time. Second, as the ionic strength of the solution is high, the presence of other cations in solution may shield cysteamine from entering the fiber and thus re- ducing disulfide bonds. In the pH range of 7.0-8.0, two cysteamine species are present in solution: 1) - S - CH 2 - CH2 - NH 3 + and 2) HS- CH• - CH• - NH 3 + (Figure 7b). The first species may either actively reduce the disulfide bonds in the hair or may form a five-membered ring in solution via hydrogen bonding. If molecular charge plays a role, we should observe a delayed reaction for ATG that might encounter a double-layer repulsion near the fiber surface because of its net negative charge. The deprotonation of ATG follows the path (Figure 7c) whereby at pH 9.4 100% of the carboxylic acid and 10% of the thiol groups are deprotonated. As the ionic strength of the solution is high, the presence of cations in solution shields the negative charges in the hair fiber and on the ATG molecules, thereby allowing ATG to diffuse into the fiber and reduce disulfide bonds. This conclusion is supported by the
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)