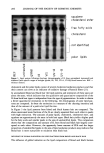
276 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS we developed a method that utilizes the unique approach of derivatizing formaldehyde with acetylacetone at room temperature, in the presence of ammonium acetate, to form the derivative 3,5-diacetyl-1,4-dihydrolutidine directly in the sample. The derivatiza- tion process is completed in 90 minutes at room temperature. The derivative is then subsequently quantitated via high-performance liquid chromatography using a reverse- phase C 18 column with water/acetonitrile (80:20) as a mobile phase. Detection is at 414 nm (5). FORMIC ACID AND ACETIC ACID Both formic and acetic acids are assayed by high-performance liquid chromatography using a polymeric polystyrene divinylbenzene sulfonate column. The mobile phase is 0.001 N aqueous sulfuric acid solution, and the UV/VIS programmable detector is set at 210 nm (6,7). PROTOCOL We investigated the kinetics of DHA in reference to pH, time, and temperature. We studied the influence of certain types of cosmetic ingredients and chemical classes of raw materials to inhibit or stabilize degradation of DHA in cosmetic products. RESULTS The analysis of 5% aqueous DHA solutions stored at room temperature showed in- creased levels of formaldehyde, formic acid, and acetic acid over time (Figure 1). If samples are stored at 45øC, increased levels of these by-products are observed, indicating a greater degradation of DHA. We also found that changes in the initial pH of aqueous solutions of DHA from acidic to alkaline lead to higher levels of formaldehyde, formic acid, and acetic acid over time. The influence of certain raw materials was also investigated in 5 % DHA solutions. We found that raw materials can affect DHA by increasing, decreasing, or having no effect on the formation of by-products and, therefore, on the degradation of DHA (Figure 2). We also applied our acquired knowledge of the interactions between DHA and certain o Figure 1. Time study of DHA degradation.
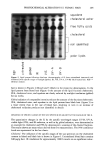
PREPRINTS OF THE 1996 ANNUAL SCIENTIFIC MEETING 277 [ l •%[]-I•'•n I •CN ] o 111v• Figure 2. Effect of cosmetic ingredients on formaldehyde formation. FORMALDEHYDE ./ 70,, / 60, so j./ 40 •-"/ • i a. :30, 20. 10 0- 1 3 9 27 36 0 14 30 49 FORMULA IN DEVELOPMENT FINAL FORMULA Time (In weeks) Figure 3. Stability of DHA degradation in new cosmetic products. types of ingredients used in cosmetic formulations and were able to stabilize dihydroxy- acetone in different types of cosmetic formulations: creams, lotions, mists and sprays (Figure 3). CONCLUSIONS As a result of our investigations and experiments via HPLC and GC/MS, we are pro- posing a mechanism for DHA degradation. Using our findings, we examined and insured the necessary conditions regarding DHA as an incoming raw material and as an ingredient in products. Such technology has resulted in a significant improvement of the stability of Estee Lauder self-tanning products. REFERENCES (1) A. A. Fisher, Contact Dermatitis (Lea & Febiger, Philadelphia, 1986). (2) W. R. Summers, Anal. Chem., 62, 1397-1402 (1990). (3) T. Nash, Biochem. J., 55, 416-421 (1953). (4) A. H. J. Gromping et al., Chromatographia, 35(3/4), 142-148 (1993).
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)