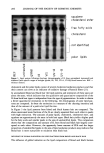
278 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS (5) A. Ostrovskaya et al., Pittsburgh Conference Abstracts, 916 (1996). (6) R. E. Smith, Ion Chromatography Applications (CRC Press, Boca Raton, FL, 1988). (7) D. P. Lee et al., LC-GC, 5, 261-266 (1986). Stabilization and delivery of vitamins and enzymes using multiple emulsions GERD H. DAHMS, IFAC (Institute for Applied Colloidtechnology), Duisberg, Germany. INTRODUCTION The ability to deliver highly specific and complex actives to the skin will become increasingly important in cosmetic and "cosmeceutical" formulations. Many of these actives will not be stable in the traditional water or oil phases of O/W or w/o emulsions. New technologies will be necessary to permit such actives to retain and deliver their biological activity on the skin. Vitamins and enzymes have gained increasing visibility in the area of skin care as antioxidants with desirable activity but often poor stability in cosmetic vehicles. We have applied our experience in the formulation of multiple emulsion systems to develop an approach to the encapsulation and controlled delivery to the skin of such ingredients. FORMULATION OF STABLE MULTIPLE EMULSIONS Multiple emulsions can be defined as X-in-oil-in-Y systems, most commonly water-in- oil-in-water systems (Figure 1). They have a long history of study as systems with significant potential for encapsulation, targeted delivery, and controlled release, but have not been widely adopted because of the traditional difficulty of stabilizing them. We have found that the viscosity and elasticity of the interfacial films seem to be particularly strong predictors of multiple emulsion stability. These parameters deter- mine the coalescence rate in the w/o and w/O/W emulsion. Rigid interfacial layers provide a barrier to coalescence. They also provide a barrier to the phase-transfer of actives. Thus the selection of emulsifiers for their capability to build liquid crystalline structures at both the w/o and O/W interfaces in multiple emulsions has been found to improve stability and encapsulation capability markedly (1). An excellent screening technique for w/o emulsifiers in this regard is the retention time of a water droplet at an oil-water interface (Figure 2). Using this technique, we have been able to select emulsifiers that permit use of a polyol (for instance propylene glycol) as the internal "aqueous" phase in place of water. Polyols have the advantage that actives
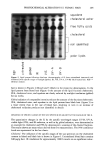
PREPRINTS OF THE 1996 ANNUAL SCIENTIFIC MEETING 279 Figure 1. Multiple emulsion schematic. Oil Phase (incl. emulsifier) Walcr Phase •'• Dinpict '• Intcffacc Figure 2. Droplet retention time method. such as vitamins and enzymes can frequently be dissolved in them without compromis- ing biological activity or stability. To encapsulate actives in multiple emulsions, a three-step procedure is necessary. First the internal w/o emulsion containing the active in the aqueous or polyol phase must be prepared. In parallel, the external aqueous phase must be prepared. Then the two must be blended. The external aqueous phase must be formulated with a rheology that ß shows excellent esthetics on the skin ß minimizes rupture and loss of encapsulated active during the blending process ß effectively stabilizes multiple droplets against creaming or coalescence once dispersed Additionally, no micelles must be present that could migrate across the internal inter- faces. These requirements can generally be met by a well-balanced gel network struc- ture, which will allow the incorporation of nearly any ratio between 1 and 30 wt% of the primary w/o or p/o emulsion. EXPERIMENTS We were able to prepare a polyol-in-oil emulsion in which vitamin C, as ascorbic acid,
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)