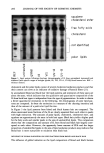
286 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS 1.0 0.$ 0.6 0.4' 0.2 ÷ 0.0. 0.0 I " ' ! " I 012 0.4 0.8 Mole F•ctlon of F• Figure 2. The vapor pressure of fragrances in laureth 4 solutions. I, PEAc. C), PEA. increase in vapor pressure relative to ideal behavior is observed until the solution is saturated at mole fraction - 1.43 X 10 -4. Vapor pressure was somewhat lower than an ideal solution when surfactant was added to phenethyl acetate until the mole fraction reached 0.8, thereafter becoming ideal (Figure 2). When a water-saturated solution was used, the vapor pressure varied with mole fraction by becoming lower than ideal solution at high fragrance concentrations and then becoming higher when the mole fraction of surfactant exceeded 0.45 (Figure 3). This is due to the fact the surfactant is in its monomeric form when the concentration is low, and at high concentrations the polar headgroups begin associating (4,5) with inverse micelies forming when water is present, such as in the saturated solution. After self-association of the surfactant occurs, a higher 0.8 0.6 0.2 0.0 o.0 0.2 0.4 0.6 018 Mole Fr•on of Fragr-aa• Figure 3. The vapor pressure of fragrances in fragrance/laureth 4 solutions saturated with water. The mole fraction is calculated on the non-aqueous compounds only. The value of Po is that of the fragrance saturated with water. I, PEAc. C), PEA.
PREPRINTS OF THE 1996 ANNUAL SCIENTIFIC MEETING 287 vapor pressure is seen because the polar groups are unavailable, and the only interactions that the fragrance molecule can experience are with the hydrocarbon chains of the surfactant, similar to the case of fragrance solubilized in decane. This also is the case for the water-saturated solution with the highest vapor pressure occurring at the point of maximum water solubilization. Low-angle x-ray diffraction shows that the fragrance molecule is located near the inter- face with the polar groups and the hydrocarbon chains and not deep within the bilayer. Its presence also influences the partitioning of water in the structure, and the Henry's constant is increased dramatically when the structure changes from liquid (1.5) to liquid crystal (12.3) (2). When comparing phenethyl acetate to phenethyl alcohol where the hydroxyl function- ality can participate, the difference in the surfactant solution is small, showing that the hydroxyl group plays only a minor role in the total molecular interaction in the water poor part of the system. In water, however, it is much more significant, and the Henry's constant is twice that for phenethyl acetate as hydrogen bonding becomes more impor- tant (3). REFERENCES (1) V. D. Roe, M. J. Lacy, and J. D. Stuart, Anal. Chem., 61, 2584 (1989). (2) S. E. Friberg, L. Fei, and P. A. Aikens, J. Mol. Liquids, submitted for publication. (3) S. E. Friberg, T. Huang, L. Fei, S. A. Vona, and P. A. Aikens, Prog. Colloid Polymer Sci., in press. (4) H. Christenson and S. E. Friberg, J. Colloid Interface Sci., 75, 276 (1980). (5) H. Christenson and S. E. Friberg, J. Phys. Chem., 84, 3633 (1980). Delivery of multi-enzyme complexes to the skin surface DAVID WATKINS, MARK ASSMANN, JENEEN WAGNER, CHARLOTTE ZHUANG, PATRICIA SEYMOUR, and JAMES HAYWARD, Col/aborative Laboratories Inc., 3 Technology Drive, East Setauket, NY 11733. Exfoliation of the dead layers of the stratum corneum in order to remove rough patches and to promote the growth of new, healthy skin is an excellent method of reducing the signs of aging. Currently products containing alpha-hydroxy acids (AHAs) are the mainstay of this treatment method. Although AHAs work well for this purpose, the low pH necessary for AHA action tends to be irritating, especially to sensitive skin, and is incompatible with many standard creams and emulsions. The search for an alternative to AHAs that avoid these problems has led us to investigate the use of enzymes as exfoliants. We examined the ability of a proteolytic enzyme to act as an exfoliant by measuring the rate of removal of the stratum corneum from human volunteers. Small patches of the volar forearm were stained for 24 hours with dihydroxy acetone, the active ingredient in
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)