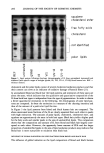
246 JOURNAL OF THE SOCIETY OF COSMETIC CHEMISTS pH 3.0, and pH 4.0, a fourth containing the placebo. Coding of the tubes maintained the double-blind design. Right and left volar forearms sites were randomized. Subjects applied a V8th inch bead of the designated lotion to each site twice daily. Sites were examined on days 1 (baseline), 7, 9, 11, 14, 16, 18, and 21. The endpoint was designated as described below. PROLONGED (CHRONIC) TREATMENT--WEEKS 5-8 During weeks 3 to 5, subjects used each lotion twice daily after the initial stained spots disappeared. At the beginning of week 5, all four sites on the forearms of all subjects were restained with dansyl chloride and the subjects resumed lotion treatments twice daily to the same designated sites until the stain disappeared the second time. ENDPOINT AND DATA ANALYSIS Because the subjects returned only every second or third day (during weekends), the exact day of stain disappearance was estimated as follows: On the day of the visit when the dansyl stain was first gone, the previous visit day number of the last score was noted and then adjusted to the "disappearance day" by adding the value of the last fluorescence score (either a 1 or 2 or 3) to the visit day number. For example, if on day 13 the score was "1" and on day 15 "0," day 14 was designated the "disappearance day." RESULTS ACUTE TREATMENT--WEEKS 1-3 The mean time for disappearance of stain was calculated for the group for each of the four treatments. The means and standard error of the mean (x + SEM) for the acute treat- ment phase (weeks 1 to 3) are shown in Figure 1. All three lactic acid-containing lotions 21 19- 16. 1'7. •6. 14. 12 II 10 1•-4 tl.O pH 2.0 15.6 .* 0.8 15.4 • 0,8 i pH 3.0 pH 4.0 Treatment Figure 1. 18.1 ! !.6 I Placebo
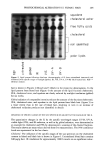
PREPRINTS OF THE 1996 ANNUAL SCIENTIFIC MEETING 247 e.l•0.7 10.1•0.e pH 2.0 pH 3.0 14.7•1.1 Treatment Figure 2. at pH 2, 3, and 4 decreased epidermal turnover time significantly compared to the placebo, with the pH 2.0 treatment showing the fastest rate of epidermal turnover. The pH 3. O-treated site was not significantly different from the pH 4.0 site, but both were different from the placebo site. Visible peeling was noted in seven of nine subjects at their pH 2.0 site, while two of nine subjects displayed peeling at their pH 3.0 site. No peeling was observed for the pH 4.0 or placebo sites. CHRONIC TREATMENT--WEEKS 5-8 The mean (+ SEM) epidermal turnover times for each of the treatments during the chronic treatment phase are shown in Figure 2. The pH 4.0 treatment did not achieve a statistically different turnover time compared to the placebo lotion. The pH 3.0 treated site did produce a significantly faster turnover time compared to both pH 4.0 and placebo. The pH 3.0 treated site did not differ significantly from the pH 2.0 site. 24 c• 20 E •. 18- •' 16. o I- 14. • 12- ILl 10- Figure 3 p=o.05 p=o.001 p=o.02 p=o.02 18.2+1.6 15.6•'-0.8 15.4•q•).8 • 13.1+1.0 12.4+1.0 pH 2.0 pH 3.0 pH 4.0 Treatment Figure 3. • • 14.7+1.1 Placebo
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)