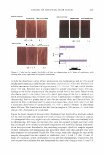
458 JOURNAL OF COSMETIC SCIENCE RESULTS Soft Quats - Gel Formers (100 g of 10% solution of anionic, titrated with 10% solution of quat) Quat Soft quats in SLS Quat Sample Soft quats in SLS-J Sample (\'iscosity of 2cl) (viscositv of e:el) 14,000 AME 7,000 13,400 DMM 6,200 DMM 6,000 MMM 50,000 Ca:MB 1,000 Ca:MB 1,000 M:MG 19,200 AMG 1,000 DMG 12,000 Miv1B 9,800 MMG 40,000 DMG 6,800 AEG 1,000 CaMG 1,000 CONCLUSIONS Quaternium compounds can be classified as hard or soft by their ability to form gelled systems with anionic systems. Cationic systems that form a gel at near stoichiometric amounts are classified as "soft", those that form precipitates of haze without appreciable viscosity build up are classified as "hard" quats. "Soft quats" can produce foam in the systems they gel, albeit at levels below the volume of foam generated by the anionic, per se. Quaternium compounds titrated with sodium laureth-3-sulfate (SLES) produced greater viscosities with amido quats. The exception was amido quats containing a benzyl group, which exhibited a low viscosity in SLES. This study was sponsored by Phoenix Chemical (Somerville N.J.) and SurfaTech Corporation (Dacula, Ga). It was conducted by Thomas O'Lenick a senior chemistry major at Georgia Southern University (synthesis) and Tim Brockman is a junior Chemistry major at Bloomsburg College (evaluation).
2005 ANNUAL SCIENTIFIC SEMINAR ALKYL POLYGLYCOSIDES DERIVATIVES Dean Smith Colonial Chemical APG compounds have been used extensively in hard surface cleaning applications, like liquid dish and liquid laundry. Recently however, the APG's have become more and more of a factor in the personal care industry. Positive Attributes of APGS I Natural - Sugar and Natural Fatty Alcohol I EO I PO Free I Biodegradable Very low toxicity I Renewable resource I Low order of skin and eye irritation I High electrolyte tolerance I "Eco Friendly" 459 How can we leverage these desirable properties to expand the use of APG into the cosmetic arena? (1) By blending them with other surfactants, or (2) by making derivatives. We have chosen the later route, and will present some of the data here. Cationic Derivatives CH3 APG-CH2CH(OH)CH2-W-R' er CH3 Cationic Products
Purchased for the exclusive use of nofirst nolast (unknown) From: SCC Media Library & Resource Center (library.scconline.org)