646 JOURNAL OF COSMETIC SCIENCE
as well as the ability to increase skin hydration and elasticity.132 Tyndallised L. acidophilus IDCC
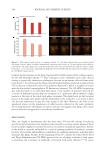
3302 restored collagen expression and decreased inflammation caused by UV exposure by
suppressing the mitogen-activated protein kinase (MAPK) signaling pathway.133 Additionally,
L. sakei lipoteichoic acid (sLTA) can inhibit the phosphorylation of MAPK, reducing matrix
metalloproteinase-1 (MMP-1) synthesis and protecting against photoaging.134
Although the literature specifically addressing the role of oral prebiotic supplementation
in skin health is limited, their complementary actions with probiotics support their use.
Moreover, prebiotic supplementation has demonstrated effective persistence throughout the
gut.135 The most widely used prebiotics include oligosaccharides (glycans), fructans (inulin-
type), sugar alcohols and complex polysaccharides (e.g., β-glucans and cellulose).136 Fructo-
oligosaccharides have been shown to reduce skin inflammation in conditions such as eczema
(also known as allergic contact dermatitis (ACD)) in mice, by increasing the abundance of B.
pseudolongum.137 Furthermore, prebiotic konjac glucomannan hydrolysates (GMH) have been
shown to inhibit Artemisia vulgaris and C. acnes by promoting the proliferation of Lactobacilli
and other microorganisms.138 In addition, heat-killed cells of L. lactis strain H61 improved skin
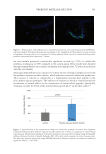
elasticity.139 Lastly, an extracellular metabolite from B. coagulans MTCC 5856 demonstrated
the ability to restore and improve skin barrier integrity, attributed to its antioxidant activity
and its ability to inhibit enzymes that degrade the extracellular matrix.140
Two dietary supplements widely advertised as nutricosmetic supplements to enhance overall
skin health are collagen and omega-3 fatty acids. Hydrolyzed collagen (HC) functions
as an antioxidant, with oral consumption increasing the abundance of collagen-derived
peptides in the bloodstream, ultimately improving skin elasticity, moisture and reducing
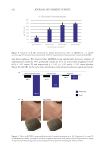
transepidermal water loss.141,142 HC has been shown to successfully diminish signs of aging,
including improvements in wrinkles and fine lines.143,144 Through oral consumption, HC
promotes fibroblast growth and collagen production in the dermis by decreasing matrix
metalloproteinase expression, which is responsible for collagen breakdown.141 A study on
a nutricosmetic blend including HC demonstrated improved structure and stratification
of consumers’ epidermal layers.145 It is worth noting that in addition to HC, this product
also contained ingredients such as hyaluronic acid, N-acetylglucosamine, borage oil,
various vitamins, minerals, antioxidants and additional bioactive ingredients. While
topical applications of HC have been explored, research suggests that oral supplementation
provides greater benefits to skin health.141 The effects of HC on the gut microbiome are
well-documented, suggesting that changes induced by HC supplementation may influence
subsequent effects on the skin.146–148
Similarly, oral supplementation with omega-3 fatty acids in the form of fish oil has been
extensively studied for its benefits to overall skin health for the past 50 years.149 Fish
oil is rich in PUFAs, particularly omega-3 fatty acids like docosahexaenoic acid (DHA)
and eicosapentaenoic acid (EPA).149 Supplementation with fish oil/omega-3 has been
associated with improvements in skin conditions like photoaging, skin cancer, dermatitis
and cutaneous wounds, as well as changes to the gut microbiome, suggesting a potential
mechanism of influence.150–161
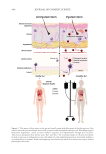
EMERGING EVIDENCE OF THE SKIN-GUT AXIS AND ITS IMPLICATIONS FOR
HUMAN HEALTH
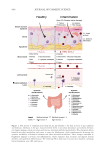
The expanding applications of the gut-skin axis reveal emerging evidence supporting the
potentially bidirectional nature of this communication pathway. While it is well-established
that the gut microbiome influences various organs such as the lungs, heart, brain and skin,
as well as the ability to increase skin hydration and elasticity.132 Tyndallised L. acidophilus IDCC
3302 restored collagen expression and decreased inflammation caused by UV exposure by
suppressing the mitogen-activated protein kinase (MAPK) signaling pathway.133 Additionally,
L. sakei lipoteichoic acid (sLTA) can inhibit the phosphorylation of MAPK, reducing matrix
metalloproteinase-1 (MMP-1) synthesis and protecting against photoaging.134
Although the literature specifically addressing the role of oral prebiotic supplementation
in skin health is limited, their complementary actions with probiotics support their use.
Moreover, prebiotic supplementation has demonstrated effective persistence throughout the
gut.135 The most widely used prebiotics include oligosaccharides (glycans), fructans (inulin-
type), sugar alcohols and complex polysaccharides (e.g., β-glucans and cellulose).136 Fructo-
oligosaccharides have been shown to reduce skin inflammation in conditions such as eczema
(also known as allergic contact dermatitis (ACD)) in mice, by increasing the abundance of B.
pseudolongum.137 Furthermore, prebiotic konjac glucomannan hydrolysates (GMH) have been
shown to inhibit Artemisia vulgaris and C. acnes by promoting the proliferation of Lactobacilli
and other microorganisms.138 In addition, heat-killed cells of L. lactis strain H61 improved skin
elasticity.139 Lastly, an extracellular metabolite from B. coagulans MTCC 5856 demonstrated
the ability to restore and improve skin barrier integrity, attributed to its antioxidant activity
and its ability to inhibit enzymes that degrade the extracellular matrix.140
Two dietary supplements widely advertised as nutricosmetic supplements to enhance overall
skin health are collagen and omega-3 fatty acids. Hydrolyzed collagen (HC) functions
as an antioxidant, with oral consumption increasing the abundance of collagen-derived
peptides in the bloodstream, ultimately improving skin elasticity, moisture and reducing
transepidermal water loss.141,142 HC has been shown to successfully diminish signs of aging,
including improvements in wrinkles and fine lines.143,144 Through oral consumption, HC
promotes fibroblast growth and collagen production in the dermis by decreasing matrix
metalloproteinase expression, which is responsible for collagen breakdown.141 A study on
a nutricosmetic blend including HC demonstrated improved structure and stratification
of consumers’ epidermal layers.145 It is worth noting that in addition to HC, this product
also contained ingredients such as hyaluronic acid, N-acetylglucosamine, borage oil,
various vitamins, minerals, antioxidants and additional bioactive ingredients. While
topical applications of HC have been explored, research suggests that oral supplementation
provides greater benefits to skin health.141 The effects of HC on the gut microbiome are
well-documented, suggesting that changes induced by HC supplementation may influence
subsequent effects on the skin.146–148
Similarly, oral supplementation with omega-3 fatty acids in the form of fish oil has been
extensively studied for its benefits to overall skin health for the past 50 years.149 Fish
oil is rich in PUFAs, particularly omega-3 fatty acids like docosahexaenoic acid (DHA)
and eicosapentaenoic acid (EPA).149 Supplementation with fish oil/omega-3 has been
associated with improvements in skin conditions like photoaging, skin cancer, dermatitis
and cutaneous wounds, as well as changes to the gut microbiome, suggesting a potential
mechanism of influence.150–161
EMERGING EVIDENCE OF THE SKIN-GUT AXIS AND ITS IMPLICATIONS FOR
HUMAN HEALTH
The expanding applications of the gut-skin axis reveal emerging evidence supporting the
potentially bidirectional nature of this communication pathway. While it is well-established
that the gut microbiome influences various organs such as the lungs, heart, brain and skin,